InVivoMAb anti-mouse CXCR3 (CD183)
Product Details
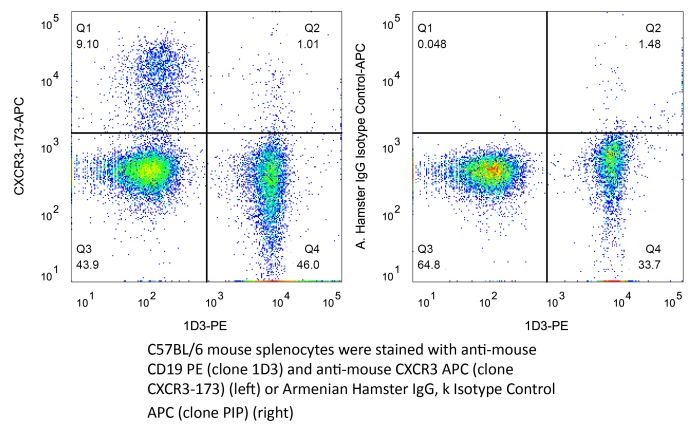
The CXCR3-173 monoclonal antibody reacts with mouse CXCR3 also known as CD183, a 38 kDa chemokine receptor for CXCL9 (MIG), CXCL10 (IP-10), and CXCL11 (ITAC). CXCR3 is expressed primarily on activated T cells, NK cells, as well as some epithelial cells and endothelial cells. CXCR3 mediates leukocyte trafficking. Binding of chemokine ligands to CXCR3 induces various cellular responses, including integrin activation, cytoskeletal changes and chemotactic migration. The binding of CXCR3-173 to CXCR3 is reported to inhibit receptor binding of CXCL10 and CXCL11 but not CXCL9.Specifications
| Isotype | Armenian hamster IgG |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb polyclonal Armenian hamster IgG |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Peptide consisting of amino acids 1-37 of mouse CXCR3 |
| Reported Applications |
in vivo CXCR3 neutralization Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_2687730 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vivo CXCR3 neutralization
Jacquelot, N., et al. (2016). "Chemokine receptor patterns in lymphocytes mirror metastatic spreading in melanoma" J Clin Invest 126(3): 921-937. PubMed
Melanoma prognosis is dictated by tumor-infiltrating lymphocytes, the migratory and functional behavior of which is guided by chemokine or cytokine gradients. Here, we retrospectively analyzed the expression patterns of 9 homing receptors (CCR/CXCR) in naive and memory CD4+ and CD8+ T lymphocytes in 57 patients with metastatic melanoma (MMel) with various sites of metastases to evaluate whether T cell CCR/CXCR expression correlates with intratumoral accumulation, metastatic progression, and/or overall survival (OS). Homing receptor expression on lymphocytes strongly correlated with MMel dissemination. Loss of CCR6 or CXCR3, but not cutaneous lymphocyte antigen (CLA), on circulating T cell subsets was associated with skin or lymph node metastases, loss of CXCR4, CXCR5, and CCR9 corresponded with lung involvement, and a rise in CCR10 or CD103 was associated with widespread dissemination. High frequencies of CD8+CCR9+ naive T cells correlated with prolonged OS, while neutralizing the CCR9/CCL25 axis in mice stimulated tumor progression. The expansion of CLA-expressing effector memory CD8+ T cells in response to a single administration of CTLA4 blockade predicted disease control at 3 months in 47 patients with MMel. Thus, specific CCR/CXCR expression patterns on circulating T lymphocytes may guide potential diagnostic and therapeutic approaches.
in vivo CXCR3 neutralization, Flow Cytometry
Chaturvedi, V., et al. (2015). "CXCR3 blockade protects against Listeria monocytogenes infection-induced fetal wastage" J Clin Invest 125(4): 1713-1725. PubMed
Mammalian pregnancy requires protection against immunological rejection of the developing fetus bearing discordant paternal antigens. Immune evasion in this developmental context entails silenced expression of chemoattractant proteins (chemokines), thereby preventing harmful immune cells from penetrating the maternal-fetal interface. Here, we demonstrate that fetal wastage triggered by prenatal Listeria monocytogenes infection is driven by placental recruitment of CXCL9-producing inflammatory neutrophils and macrophages that promote infiltration of fetal-specific T cells into the decidua. Maternal CD8+ T cells with fetal specificity upregulated expression of the chemokine receptor CXCR3 and, together with neutrophils and macrophages, were essential for L. monocytogenes-induced fetal resorption. Conversely, decidual accumulation of maternal T cells with fetal specificity and fetal wastage were extinguished by CXCR3 blockade or in CXCR3-deficient mice. Remarkably, protection against fetal wastage and in utero L. monocytogenes invasion was maintained even when CXCR3 neutralization was initiated after infection, and this protective effect extended to fetal resorption triggered by partial ablation of immune-suppressive maternal Tregs, which expand during pregnancy to sustain fetal tolerance. Together, our results indicate that functionally overriding chemokine silencing at the maternal-fetal interface promotes the pathogenesis of prenatal infection and suggest that therapeutically reinforcing this pathway represents a universal approach for mitigating immune-mediated pregnancy complications.
in vivo CXCR3 neutralization
Glennie, N. D., et al. (2015). "Skin-resident memory CD4+ T cells enhance protection against Leishmania major infection" J Exp Med 212(9): 1405-1414. PubMed
Leishmaniasis causes a significant disease burden worldwide. Although Leishmania-infected patients become refractory to reinfection after disease resolution, effective immune protection has not yet been achieved by human vaccines. Although circulating Leishmania-specific T cells are known to play a critical role in immunity, the role of memory T cells present in peripheral tissues has not been explored. Here, we identify a population of skin-resident Leishmania-specific memory CD4(+) T cells. These cells produce IFN-gamma and remain resident in the skin when transplanted by skin graft onto naive mice. They function to recruit circulating T cells to the skin in a CXCR3-dependent manner, resulting in better control of the parasites. Our findings are the first to demonstrate that CD4(+) TRM cells form in response to a parasitic infection, and indicate that optimal protective immunity to Leishmania, and thus the success of a vaccine, may depend on generating both circulating and skin-resident memory T cells.
in vivo CXCR3 neutralization
Yang, H., et al. (2015). "STAT3 Inhibition Enhances the Therapeutic Efficacy of Immunogenic Chemotherapy by Stimulating Type 1 Interferon Production by Cancer Cells" Cancer Res 75(18): 3812-3822. PubMed
STAT3 is an oncogenic transcription factor with potent immunosuppressive functions. We found that pharmacologic inhibition of STAT3 or its selective knockout in cancer cells improved the tumor growth-inhibitory efficacy of anthracycline-based chemotherapies. This combined effect of STAT3 inhibition/depletion and anthracyclines was only found in tumors growing on immunocompetent (not in immunodeficient) mice. As compared with Stat3-sufficient control tumors, Stat3(-/-) cancer cells exhibited an increased infiltration by dendritic cells and cytotoxic T lymphocytes after chemotherapy. Anthracyclines are known to induce several stress pathways that enhance the immunogenicity of dying and dead cancer cells, thereby stimulating a dendritic cell-dependent and T lymphocyte-mediated anticancer immune response. Among these therapy-relevant stress pathways, Stat3(-/-) cancer cells manifested one significant improvement, namely an increase in the expression of multiple type-1 interferon-responsive genes, including that of the chemokines Cxcl9 and Cxcl10. This enhanced type-1 interferon response could be suppressed by reintroducing wild-type Stat3 (but not a transactivation-deficient mutant Stat3(Y705F)) into the tumor cells. This maneuver also abolished the improved chemotherapeutic response of Stat3(-/-) cancers. Finally, the neutralization of the common type-1 interferon receptor or that of the chemokine receptor CXCR3 (which binds CXCL9 and CXCL10) abolished the difference in the chemotherapeutic response between Stat3(-/-) and control tumors. Altogether, these results suggest that STAT3 inhibitors may improve the outcome of chemotherapy by enhancing the type-1 interferon response of cancer cells.