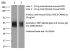
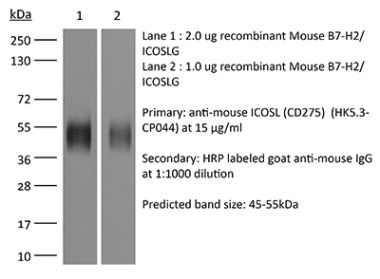
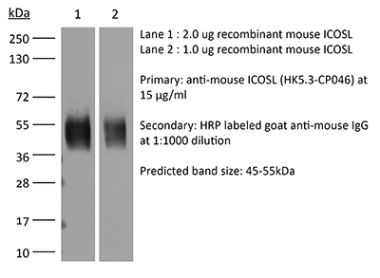
InVivoMAb anti-mouse ICOSL (CD275)
Product Details
The HK5.3 monoclonal antibody reacts with mouse ICOSL (inducible T cell co-stimulator ligand) also known as CD275, B7RP-1, and B7-H2. ICOSL is a 40 kDa immune checkpoint protein belonging to the Ig receptor superfamily. Upon ICOSL binding, ICOS signaling co-stimulates T and B cell responses. The ligand Is expressed on antigen presenting cells including splenic B cells, dendritic cells, and macrophages. ICOS signaling is also thought to be important for maintaining regulatory T cell homeostasis. The HK5.3 antibody has been shown to block the binding of ICOSL to ICOS both in vitroand in vivo. HK5.3 treatment of mice has been reported to lead to a loss of regulatory T cells.Specifications
| Isotype | Rat IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG2a isotype control, anti-trinitrophenol |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse B7-RP1 transfected cell line |
| Reported Applications | in vivo ICOSL neutralization |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107566 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Additional Formats
Recommended Products
in vivo ICOSL neutralization
Stone, E. L., et al. (2015). "ICOS coreceptor signaling inactivates the transcription factor FOXO1 to promote Tfh cell differentiation" Immunity 42(2): 239-251. PubMed
T follicular helper (Tfh) cells are essential in the induction of high-affinity, class-switched antibodies. The differentiation of Tfh cells is a multi-step process that depends upon the co-receptor ICOS and the activation of phosphoinositide-3 kinase leading to the expression of key Tfh cell genes. We report that ICOS signaling inactivates the transcription factor FOXO1, and a Foxo1 genetic deletion allowed for generation of Tfh cells with reduced dependence on ICOS ligand. Conversely, enforced nuclear localization of FOXO1 inhibited Tfh cell development even though ICOS was overexpressed. FOXO1 regulated Tfh cell differentiation through a broad program of gene expression exemplified by its negative regulation of Bcl6. Final differentiation to germinal center Tfh cells (GC-Tfh) was instead FOXO1 dependent as the Foxo1(-/-) GC-Tfh cell population was substantially reduced. We propose that ICOS signaling transiently inactivates FOXO1 to initiate a Tfh cell contingency that is completed in a FOXO1-dependent manner.
in vivo ICOSL neutralization
Raynor, J., et al. (2015). "IL-6 and ICOS Antagonize Bim and Promote Regulatory T Cell Accrual with Age" J Immunol 195(3): 944-952. PubMed
Regulatory T cells (Tregs), a subset of CD4(+) T cells, dramatically accumulate with age in humans and mice and contribute to age-related immune suppression. Recently, we showed that a majority of accumulating Tregs in aged mice expressed low levels of CD25, and their accrual is associated with declining levels of IL-2 in aged mice. In this study, we further investigated the origin of CD25(lo) Tregs in aged mice. First, aged Tregs had high expression of neuropilin-1 and Helios, and had a broad Vbeta repertoire. Next, we analyzed the gene expression profile of Tregs, naive T cells, and memory T cells in aged mice. We found that the gene expression profile of aged CD25(lo) Tregs were more related to young CD25(lo) Tregs than to either naive or memory T cells. Further, the gene expression profile of aged Tregs was consistent with recently described “effector” Tregs (eTregs). Additional analysis revealed that nearly all Tregs in aged mice were of an effector phenotype (CD44(hi)CD62L(lo)) and could be further characterized by high levels of ICOS and CD69. ICOS contributed to Treg maintenance in aged mice, because in vivo Ab blockade of ICOSL led to a loss of eTregs, and this loss was rescued in Bim-deficient mice. Further, serum levels of IL-6 increased with age and contributed to elevated expression of ICOS on aged Tregs. Finally, Treg accrual was significantly blunted in aged IL-6-deficient mice. Together, our data show a role for IL-6 in promoting eTreg accrual with age likely through maintenance of ICOS expression.
in vivo ICOSL neutralization
Xin, L., et al. (2014). "Commensal microbes drive intestinal inflammation by IL-17-producing CD4+ T cells through ICOSL and OX40L costimulation in the absence of B7-1 and B7-2" Proc Natl Acad Sci U S A 111(29): 10672-10677. PubMed
The costimulatory B7-1 (CD80)/B7-2 (CD86) molecules, along with T-cell receptor stimulation, together facilitate T-cell activation. This explains why in vivo B7 costimulation neutralization efficiently silences a variety of human autoimmune disorders. Paradoxically, however, B7 blockade also potently moderates accumulation of immune-suppressive regulatory T cells (Tregs) essential for protection against multiorgan systemic autoimmunity. Here we show that B7 deprivation in mice overrides the necessity for Tregs in averting systemic autoimmunity and inflammation in extraintestinal tissues, whereas peripherally induced Tregs retained in the absence of B7 selectively mitigate intestinal inflammation caused by Th17 effector CD4(+) T cells. The need for additional immune suppression in the intestine reflects commensal microbe-driven T-cell activation through the accessory costimulation molecules ICOSL and OX40L. Eradication of commensal enteric bacteria mitigates intestinal inflammation and IL-17 production triggered by Treg depletion in B7-deficient mice, whereas re-establishing intestinal colonization with Candida albicans primes expansion of Th17 cells with commensal specificity. Thus, neutralizing B7 costimulation uncovers an essential role for Tregs in selectively averting intestinal inflammation by Th17 CD4(+) T cells with commensal microbe specificity.
in vivo ICOSL neutralization
Huang, W., et al. (2014). "IL-2-inducible T cell kinase tunes T regulatory cell development and is required for suppressive function" J Immunol 193(5): 2267-2272. PubMed
IL-2-inducible T cell kinase (ITK) is a key signaling mediator downstream of TCR, mediating T cell positive selection, as well as innate T cell and CD4(+) Th2/Th17 differentiation. In this article, we show that ITK also negatively tunes IL-2-induced expansion of conventional Foxp3-expressing regulatory T cells (Tregs). In vivo, Treg abundance is inversely correlated with ITK expression, and inducible Treg development is inversely dependent on ITK kinase activity. While Treg development normally requires both hematopoietic and thymic MHC class 2 (MHC2) expression, the absence of ITK allows Treg development with MHC2 expression in either compartment, with preference for selection by thymic MHC2, suggesting a gatekeeper role for ITK in ensuring that only Tregs selected by both thymic and hematopoietic MHC2 survive selection. Although ITK suppresses Treg development and is not required for maintenance of neuropilin-1-positive natural Tregs in the periphery, it is indispensable for Treg functional suppression of naive CD4(+) T cell-induced colitis in Rag(-/-) recipients. ITK thus regulates the development and function of Tregs.
in vivo ICOSL neutralization
Srivastava, S., et al. (2014). "Type I interferons directly inhibit regulatory T cells to allow optimal antiviral T cell responses during acute LCMV infection" J Exp Med 211(5): 961-974. PubMed
Regulatory T (T reg) cells play an essential role in preventing autoimmunity but can also impair clearance of foreign pathogens. Paradoxically, signals known to promote T reg cell function are abundant during infection and could inappropriately enhance T reg cell activity. How T reg cell function is restrained during infection to allow the generation of effective antiviral responses remains largely unclear. We demonstrate that the potent antiviral type I interferons (IFNs) directly inhibit co-stimulation-dependent T reg cell activation and proliferation, both in vitro and in vivo during acute infection with lymphocytic choriomeningitis virus (LCMV). Loss of the type I IFN receptor specifically in T reg cells results in functional impairment of virus-specific CD8(+) and CD4(+) T cells and inefficient viral clearance. Together, these data demonstrate that inhibition of T reg cells by IFNs is necessary for the generation of optimal antiviral T cell responses during acute LCMV infection.
in vivo ICOSL neutralization
Colino, J., et al. (2013). "Noncovalent association of protein and capsular polysaccharide on bacteria-sized latex beads as a model for polysaccharide-specific humoral immunity to intact gram-positive extracellular bacteria" J Immunol 191(6): 3254-3263. PubMed
Intact Streptococcus pneumoniae expressing type 14 capsular polysaccharide (PPS14) and type III S. agalactiae containing a PPS14 core capsule identical to PPS14 exhibit noncovalent associations of PPS14 and bacterial protein, in contrast to soluble covalent conjugates of these respective Ags. Both bacteria and conjugates induce murine PPS14-specific IgG responses dependent on CD4(+) T cells. Further, secondary immunization with conjugate and S. agalactiae, although not S. pneumoniae, results in a boosted response. However, in contrast to conjugate, PPS14-specific IgG responses to bacteria lack affinity maturation use the 44.1-idiotype and are dependent on marginal zone B cells. To better understand the mechanism underlying this dichotomy, we developed a minimal model of intact bacteria in which PPS14 and pneumococcal surface protein A (PspA) were stably attached to 1 mum (bacteria-sized) latex beads, but not directly linked to each other, in contrast to PPS14-PspA conjugate. Beads coated simultaneously with PPS14+, similar to conjugate, induced in mice boosted PPS14-specific IgG secondary responses, dependent on T cells and ICOS-dependent costimulation, and in which priming could be achieved with PspA alone. In contrast to conjugate, but similar to intact bacteria, the primary PPS14-specific IgG response to beads coated simultaneously with PPS14+ peaked rapidly, with the secondary response highly enriched for the 44.1-idiotype and lacking affinity maturation. These results demonstrate that noncovalent association in a particle, of polysaccharide and protein, recapitulates essential immunologic characteristics of intact bacteria that are distinct from soluble covalent conjugates of these respective Ags.
in vivo ICOSL neutralization
Baumjohann, D., et al. (2013). "Persistent antigen and germinal center B cells sustain T follicular helper cell responses and phenotype" Immunity 38(3): 596-605. PubMed
T follicular helper (Tfh) cells provide help to B cells and are crucial for establishment of germinal center (GC) reactions, including production of high-affinity antibodies and generation of memory B cells and long-lived plasma cells. Here we report that the magnitude of the Tfh cell response was dictated by the amount of antigen and directly correlated with the magnitude of the GC B cell response. In addition, maintenance of the Tfh cell phenotype required sustained antigenic stimulation by GC B cells. In lymphopenic conditions, a strong and prolonged Tfh cell response led to bystander B cell activation, hypergammaglobulinemia, and production of poly- and self-reactive antibodies. These data demonstrate that antigen dose determines the size and duration of the Tfh cell response and GC reaction, highlight the transient nature of the Tfh cell phenotype, and suggest a link between overstimulation of Tfh cells and the development of dysregulated humoral immune responses.
in vivo ICOSL neutralization
Arjunaraja, S., et al. (2012). "Structurally identical capsular polysaccharide expressed by intact group B streptococcus versus Streptococcus pneumoniae elicits distinct murine polysaccharide-specific IgG responses in vivo" J Immunol 188(11): 5238-5246. PubMed
We previously reported distinct differences in the murine in vivo Ig polysaccharide (PS)-specific responses to intact Streptococcus pneumoniae compared with responses to Neisseria meningitidis and that in each case, the bacterial subcapsular domain markedly influences the Ig response to the associated PS. In light of potentially unique contributions of biochemically distinct capsular PS and/or their characteristic attachments to the underlying bacterium, it remains unresolved whether different bacterial subcapsular domains can exert differential effects on PS-specific Ig responses to distinct bacterial pathogens. In this report, we used a mutant strain of group B Streptococcus (Streptococcus agalactiae) type III (GBS-III) that expresses desialylated capsular polysaccharide of GBS-III, biochemically identical to capsular pneumococcal polysaccharide type 14 (PPS14) of Streptococcus pneumoniae (intact inactivated Streptococcus pneumoniae, capsular type 14, Pn14), directly to compare the in vivo PPS14-specific IgG responses to two distinct gram-positive bacteria. Although both GBS-III and Pn14 elicited relatively rapid primary PPS14-specific IgG responses dependent on CD4(+) T cells, B7-dependent costimulation, and CD40-CD40L interactions, only GBS-III induced a highly boosted ICOS-dependent PPS14-specific IgG response after secondary immunization. Of note, priming with Pn14 and boosting with GBS-III, although not isolated PPS14, elicited a similar boosted PPS14-specific IgG response that was dependent on CD4(+) T cells during secondary immunization, indicating that Pn14 primes for memory but, unlike GBS-III, fails to elicit it. The inability of Pn14 to elicit a boosted PPS14-specific IgG response was overcome by coimmunization with unencapsulated GBS-III. Collectively, these data establish that structurally identical capsular PS expressed by two distinct gram-positive extracellular bacteria can indeed elicit distinct PS-specific IgG responses in vivo.
in vivo ICOSL neutralization
Arjunaraja, S., et al. (2012). "The nature of an in vivo anti-capsular polysaccharide response is markedly influenced by the composition and/or architecture of the bacterial subcapsular domain" J Immunol 188(2): 569-577. PubMed
In vivo anti-polysaccharide Ig responses to isolated polysaccharide (PS) are T cell independent, rapid, and fail to generate memory. However, little is known regarding PS-specific Ig responses to intact gram-positive and gram-negative extracellular bacteria. We previously demonstrated that intact heat-killed Streptococcus pneumoniae, a gram-positive bacterium, elicited a rapid primary pneumococcal capsular PS (PPS) response in mice that was dependent on CD4(+) T cells, B7-dependent costimulation, and CD40-CD40L interactions. However, this response was ICOS independent and failed to generate a boosted PPS-specific secondary IgG response. In the current study, we analyzed the murine meningococcal type C PS (MCPS)-specific Ig response to i.p.-injected intact, heat-killed Neisseria meningitidis, serogroup C (MenC), a gram-negative bacterium. In contrast to S. pneumoniae, the IgG anti-MCPS response to MenC exhibited delayed primary kinetics and was highly boosted after secondary immunization, whereas the IgG anti-MCPS response to isolated MCPS was rapid, without secondary boosting, and consisted of only IgG1 and IgG3, as opposed to all four IgG isotypes in response to intact MenC. The secondary, but not primary, IgG anti-MCPS response to MenC was dependent on CD4(+) T cells, CD40L, CD28, and ICOS. The primary and secondary IgG anti-MCPS responses were lower in TLR4-defective (C3H/HeJ) but not TLR2(-/-) or MyD88(-/-) mice, but secondary boosting was still observed. Of interest, coimmunization of S. pneumoniae and MenC resulted in a boosted secondary IgG anti-PPS response to S. pneumoniae. Our data demonstrate that the nature of the in vivo anti-PS response is markedly influenced by the composition and/or architecture of the bacterial subcapsular domain.
in vivo ICOSL neutralization
Choi, Y. S., et al. (2011). "ICOS receptor instructs T follicular helper cell versus effector cell differentiation via induction of the transcriptional repressor Bcl6" Immunity 34(6): 932-946. PubMed
The nature of follicular helper CD4(+) T (Tfh) cell differentiation remains controversial, including the minimal signals required for Tfh cell differentiation and the time at which Tfh cell differentiation occurs. Here we determine that Tfh cell development initiates immediately during dendritic cell (DC) priming in vivo. We demonstrate that inducible costimulator (ICOS) provides a critical early signal to induce the transcription factor Bcl6, and Bcl6 then induces CXCR5, the canonical feature of Tfh cells. Strikingly, a bifurcation between Tfh and effector Th cells was measurable by the second cell division of CD4(+) T cells, at day 2 after an acute viral infection: IL2Ralpha(int) cells expressed Bcl6 and CXCR5 (Tfh cell program), whereas IL2Ralpha(hi) cells exhibited strong Blimp1 expression that repressed Bcl6 (effector Th cell program). Virtually complete polarization between Bcl6(+) Tfh cells and Blimp1(+) effector Th cell populations developed by 72 hr, even without B cells. Tfh cells were subsequently lost in the absence of B cells, demonstrating a B cell requirement for maintenance of Bcl6 and Tfh cell commitment via sequential ICOS signals.