InVivoMAb anti-mouse CD1d (CD1.1)
Product Details
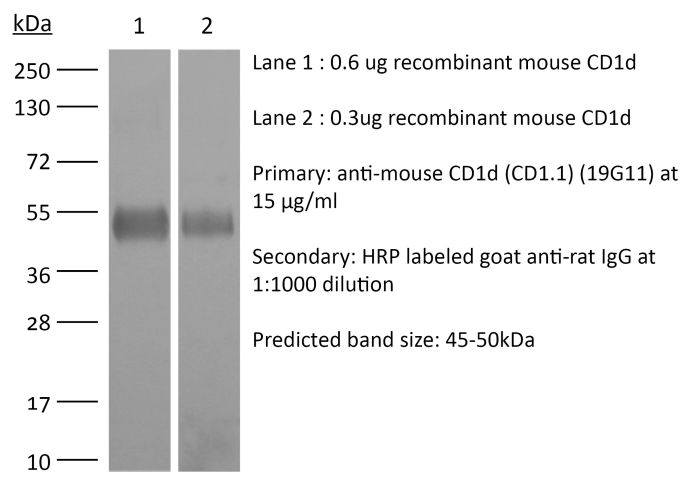
The 19G11 monoclonal antibody reacts with mouse CD1d also known as CD1.1. CD1d is a 48 kDa type I membrane glycoprotein and a member of the CD1 family of glycoproteins. CD1d is a non-classical MHC protein with structural homology to class I MHC molecules. CD1d is expressed on the surface of various antigen-presenting cells and is involved in the presentation of non-peptide glycolipid antigens to to CD1d-restricted T cells. CD1d-presented glycolipid antigens activate invariant natural killer T (iNKT) cells, through the interaction with the T-cell receptor present on iNKT cell membranes. When activated, iNKT cells rapidly produce Th1 and Th2 cytokines.Specifications
| Isotype | Rat IgG1 |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG1 isotype control, anti-horseradish peroxidase |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse CD1d |
| Reported Applications |
in vivo CD1d neutralization in vitro CD1d neutralization |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107568 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vitro CD1d neutralization
Melum, E., et al. (2019). "Control of CD1d-restricted antigen presentation and inflammation by sphingomyelin" Nat Immunol 20(12): 1644-1655. PubMed
Invariant natural killer T (iNKT) cells recognize activating self and microbial lipids presented by CD1d. CD1d can also bind non-activating lipids, such as sphingomyelin. We hypothesized that these serve as endogenous regulators and investigated humans and mice deficient in acid sphingomyelinase (ASM), an enzyme that degrades sphingomyelin. We show that ASM absence in mice leads to diminished CD1d-restricted antigen presentation and iNKT cell selection in the thymus, resulting in decreased iNKT cell levels and resistance to iNKT cell-mediated inflammatory conditions. Defective antigen presentation and decreased iNKT cells are also observed in ASM-deficient humans with Niemann-Pick disease, and ASM activity in healthy humans correlates with iNKT cell phenotype. Pharmacological ASM administration facilitates antigen presentation and restores the levels of iNKT cells in ASM-deficient mice. Together, these results demonstrate that control of non-agonistic CD1d-associated lipids is critical for iNKT cell development and function in vivo and represents a tight link between cellular sphingolipid metabolism and immunity.
in vivo CD1d neutralization
Olszak, T., et al. (2014). "Protective mucosal immunity mediated by epithelial CD1d and IL-10" Nature 509(7501): 497-502. PubMed
The mechanisms by which mucosal homeostasis is maintained are of central importance to inflammatory bowel disease. Critical to these processes is the intestinal epithelial cell (IEC), which regulates immune responses at the interface between the commensal microbiota and the host. CD1d presents self and microbial lipid antigens to natural killer T (NKT) cells, which are involved in the pathogenesis of colitis in animal models and human inflammatory bowel disease. As CD1d crosslinking on model IECs results in the production of the important regulatory cytokine interleukin (IL)-10 (ref. 9), decreased epithelial CD1d expression–as observed in inflammatory bowel disease–may contribute substantially to intestinal inflammation. Here we show in mice that whereas bone-marrow-derived CD1d signals contribute to NKT-cell-mediated intestinal inflammation, engagement of epithelial CD1d elicits protective effects through the activation of STAT3 and STAT3-dependent transcription of IL-10, heat shock protein 110 (HSP110; also known as HSP105), and CD1d itself. All of these epithelial elements are critically involved in controlling CD1d-mediated intestinal inflammation. This is demonstrated by severe NKT-cell-mediated colitis upon IEC-specific deletion of IL-10, CD1d, and its critical regulator microsomal triglyceride transfer protein (MTP), as well as deletion of HSP110 in the radioresistant compartment. Our studies thus uncover a novel pathway of IEC-dependent regulation of mucosal homeostasis and highlight a critical role of IL-10 in the intestinal epithelium, with broad implications for diseases such as inflammatory bowel disease.
in vivo CD1d neutralization
An, D., et al. (2014). "Sphingolipids from a symbiotic microbe regulate homeostasis of host intestinal natural killer T cells" Cell 156(1-2): 123-133. PubMed
Coevolution of beneficial microorganisms with the mammalian intestine fundamentally shapes mammalian physiology. Here, we report that the intestinal microbe Bacteroides fragilis modifies the homeostasis of host invariant natural killer T (iNKT) cells by supplementing the host’s endogenous lipid antigen milieu with unique inhibitory sphingolipids. The process occurs early in life and effectively impedes iNKT cell proliferation during neonatal development. Consequently, total colonic iNKT cell numbers are restricted into adulthood, and hosts are protected against experimental iNKT cell-mediated, oxazolone-induced colitis. In studies with neonatal mice lacking access to bacterial sphingolipids, we found that treatment with B. fragilis glycosphingolipids-exemplified by an isolated peak (MW = 717.6) called GSL-Bf717-reduces colonic iNKT cell numbers and confers protection against oxazolone-induced colitis in adulthood. Our results suggest that the distinctive inhibitory capacity of GSL-Bf717 and similar molecules may prove useful in the treatment of autoimmune and allergic disorders in which iNKT cell activation is destructive.