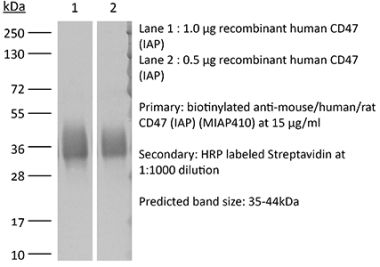
InVivoMAb anti-mouse CD47 (IAP)
Product Details
The MIAP301 monoclonal antibody reacts with mouse CD47 otherwise known as integrin-associated protein (IAP). CD47 is an approximately 50 kDa glycosylated five transmembrane protein that is ubiquitously expressed by both hematopoietic cells such as T and B lymphocytes, monocytes, platelets and erythrocytes and non-hematopoietic cells. CD47 is involved in a range of cellular processes, including apoptosis, proliferation, adhesion, and migration. Furthermore, it plays a key role in immune and angiogenic responses. CD47 is a receptor for thrombospondin-1 (TSP-1), a secreted glycoprotein that plays a role in vascular development and angiogenesis. CD47 Is has been found to be overexpressed in many different tumor cells. Because of this, anti-CD47 monoclonal antibodies have been proposed and studied as a therapeutic treatment for human cancers. The MIAP301 antibody has been shown to neutralize CD47 in vivo and in vitro.Specifications
| Isotype | Rat IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG2a isotype control, anti-trinitrophenol |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Intact CD47 purified from placenta |
| Reported Applications |
in vivo CD47 blockade in vitro CD47 blockade Immunofluorescence |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_2687793 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vivo CD47 blockade, in vitro CD47 blockade
Reed, M., et al. (2019). "Epithelial CD47 is critical for mucosal repair in the murine intestine in vivo" Nat Commun 10(1): 5004. PubMed
CD47 is a ubiquitously expressed transmembrane glycoprotein that regulates inflammatory responses and tissue repair. Here, we show that normal mice treated with anti-CD47 antibodies, and Cd47-null mice have impaired intestinal mucosal wound healing. Furthermore, intestinal epithelial cell (IEC)-specific loss of CD47 does not induce spontaneous immune-mediated intestinal barrier disruption but results in defective mucosal repair after biopsy-induced colonic wounding or Dextran Sulfate Sodium (DSS)-induced mucosal damage. In vitro analyses using primary cultures of CD47-deficient murine colonic IEC or human colonoid-derived IEC treated with CD47-blocking antibodies demonstrate impaired epithelial cell migration in wound healing assays. Defective wound repair after CD47 loss is linked to decreased epithelial β1 integrin and focal adhesion signaling, as well as reduced thrombospondin-1 and TGF-β1. These results demonstrate a critical role for IEC-expressed CD47 in regulating mucosal repair and raise important considerations for possible alterations in wound healing secondary to therapeutic targeting of CD47.
in vitro CD47 blockade
Liu, M., et al. (2019). "Metabolic rewiring of macrophages by CpG potentiates clearance of cancer cells and overcomes tumor-expressed CD47-mediated ‘don’t-eat-me’ signal" Nat Immunol 20(3): 265-275. PubMed
Macrophages enforce antitumor immunity by engulfing and killing tumor cells. Although these functions are determined by a balance of stimulatory and inhibitory signals, the role of macrophage metabolism is unknown. Here, we study the capacity of macrophages to circumvent inhibitory activity mediated by CD47 on cancer cells. We show that stimulation with a CpG oligodeoxynucleotide, a Toll-like receptor 9 agonist, evokes changes in the central carbon metabolism of macrophages that enable antitumor activity, including engulfment of CD47(+) cancer cells. CpG activation engenders a metabolic state that requires fatty acid oxidation and shunting of tricarboxylic acid cycle intermediates for de novo lipid biosynthesis. This integration of metabolic inputs is underpinned by carnitine palmitoyltransferase 1A and adenosine tri-phosphate citrate lyase, which, together, impart macrophages with antitumor potential capable of overcoming inhibitory CD47 on cancer cells. Our findings identify central carbon metabolism to be a novel determinant and potential therapeutic target for stimulating antitumor activity by macrophages.
in vivo CD47 blockade
Pan, Y., et al. (2019). "Single-cell RNA sequencing reveals compartmental remodeling of tumor-infiltrating immune cells induced by anti-CD47 targeting in pancreatic cancer" J Hematol Oncol 12(1): 124. PubMed
BACKGROUND: Human pancreatic ductal adenocarcinoma (PDAC) responds poorly to immune checkpoint inhibitor (ICPi). While the mechanism is not completely clear, it has been recognized that tumor microenvironment (TME) plays key roles. We investigated if targeting CD47 with a monoclonal antibody could enhance the response of PDAC to ICPi by altering the TME. METHODS: Using immunohistochemistry, we examined tumor-infiltrating CD68(+) pan-macrophages (CD68(+) M) and CD163(+) M2 macrophages (CD163(+) M2) and tumor expression of CD47 and PD-L1 proteins in 106 cases of PDAC. The efficacy of CD47 blockade was examined in xenograft models. CD45(+) immune cells from syngeneic tumor models were subjected to single-cell RNA-sequencing (scRNA-seq) by using the 10x Genomics pipeline. RESULTS: We found that CD47 expression correlated with the level of CD68(+) M but not CD163(+) M2. High levels of tumor-infiltrating CD68(+) M, CD163(+) M2, and CD47 expression were significantly associated with worse survival. CD47(high)/CD68(+) M(high) and CD47(high)/CD163(+) M2(high) correlated significantly with shorter survival, whereas CD47(low)/CD68(+) M(low) and CD47(low)/CD163(+) M2(low) correlated with longer survival. Intriguingly, CD47 blockade decreased the tumor burden in the Panc02 but not in the MPC-83 syngeneic mouse model. Using scRNA-seq, we showed that anti-CD47 treatment significantly remodeled the intratumoral lymphocyte and macrophage compartments in Panc02 tumor-bearing mice by increasing the pro-inflammatory macrophages that exhibit anti-tumor function, while reducing the anti-inflammatory macrophages. Moreover, CD47 blockade not only increased the number of intratumoral CD8(+) T cells, but also remodeled the T cell cluster toward a more activated one. Further, combination therapy targeting both CD47 and PD-L1 resulted in synergistic inhibition of PDAC growth in the MPC-83 but not in Panc02 model. MPC-83 but not Panc02 mice treated with both anti-CD47 and anti-PD-L1 showed increased number of PD-1(+)CD8(+) T cells and enhanced expression of key immune activating genes. CONCLUSION: Our data indicate that CD47 targeting induces compartmental remodeling of tumor-infiltrating immune cells of the TME in PDAC. Different PDAC mouse models exhibited differential response to the anti-CD47 and anti-PD-L1 blockade due to the differential effect of this combination treatment on the infiltrating immune cells and key immune activating genes in the TME established by the different PDAC cell lines.
in vivo CD47 blockade
Wu, L., et al. (2018). "Anti-CD47 treatment enhances anti-tumor T-cell immunity and improves immunosuppressive environment in head and neck squamous cell carcinoma" Oncoimmunology 7(4): e1397248. PubMed
Head and neck squamous cell carcinoma (HNSCC) is considered as an immunosuppressive disease, with impaired tumor-infiltrating T lymphocytes and increased suppressive immune cells. The efficacy of CD47 antibodies in immune checkpoint therapy is not clearly understood in HNSCC. In this study, human tissue microarrays and immunocompetent transgenic mouse models were used to explore the expression of CD47 and the use of CD47 antibodies in HNSCC. We identified overexpression of CD47 in HNSCC as compared with the control normal human tissue and also in HNSCC mouse models. The expression of CD47 also correlated with clinicopathological parameters as well as outcome. Furthermore, inhibition of CD47 delayed tumor growth and improved tumor microenvironment by stimulating effector T cells and decreasing suppressive immune cells and regulating the function of CD11b(+) Ly6G(+) MDSC. Our data suggest that CD47 blockade may be a potential immunotherapeutic target in human HNSCC.
in vivo CD47 blockade
Sallets, A., et al. (2018). "Enhancing immunotherapy of STING agonist for lymphoma in preclinical models" Blood Adv 2(17): 2230-2241. PubMed
Direct activation of tumor infiltrating antigen-presenting cells (APCs) by intratumoral injection of STING agonists (STINGa) leads to regression of the treated lymphoma tumor. Because STING activation induces apoptosis in lymphoma cells in vitro, we distinguished between the direct therapeutic vs the indirect immunotherapeutic properties of STINGa in vivo. Employing wild-type or STING knockout hosts bearing either wild-type or STING knockout tumor cells, we demonstrated that local tumor regression is totally dependent on STING expression by the host and is therefore immune mediated. However, distant untreated tumors are weakly affected after injection of STINGa to a single tumor site. Therefore, using the STINGa currently being tested in clinical trials, we screened for immunomodulatory agents that could synergize with the STING pathway to induce a systemic antitumor immune response and regression of distant tumors. We combined the STINGa with agents that improve APC or T-cell function. We found that modulation of both APCs and T cells can enhance control of distant lymphoma tumors by STINGa. In particular, adding an anti-GITR antibody induced lymphocyte expansion in the lymph node draining the treated site followed by increased T-cell infiltration in the distant tumor. Furthermore, more of these CD8 T cells at the distant site expressed PD-1. Therefore, blockade of PD-1 further enhanced tumor control at the distant site, leading to cure in 50% of the mice. These preclinical data provide the rationale for testing local injection of STINGa followed by agonistic anti-GITR and anti-PD-1 antibodies as immunotherapy for human lymphoma.
in vivo CD47 blockade
Xu, M. M., et al. (2017). "Dendritic Cells but Not Macrophages Sense Tumor Mitochondrial DNA for Cross-priming through Signal Regulatory Protein alpha Signaling" Immunity 47(2): 363-373 e365. PubMed
Inhibition of cytosolic DNA sensing represents a strategy that tumor cells use for immune evasion, but the underlying mechanisms are unclear. Here we have shown that CD47-signal regulatory protein alpha (SIRPalpha) axis dictates the fate of ingested DNA in DCs for immune evasion. Although macrophages were more potent in uptaking tumor DNA, increase of DNA sensing by blocking the interaction of SIRPalpha with CD47 preferentially occurred in dendritic cells (DCs) but not in macrophages. Mechanistically, CD47 blockade enabled the activation of NADPH oxidase NOX2 in DCs, which in turn inhibited phagosomal acidification and reduced the degradation of tumor mitochondrial DNA (mtDNA) in DCs. mtDNA was recognized by cyclic-GMP-AMP synthase (cGAS) in the DC cytosol, contributing to type I interferon (IFN) production and antitumor adaptive immunity. Thus, our findings have demonstrated how tumor cells inhibit innate sensing in DCs and suggested that the CD47-SIRPalpha axis is critical for DC-driven antitumor immunity.
in vivo CD47 blockade
Liu, X., et al. (2015). "CD47 blockade triggers T cell-mediated destruction of immunogenic tumors" Nat Med 21(10): 1209-1215. PubMed
Macrophage phagocytosis of tumor cells mediated by CD47-specific blocking antibodies has been proposed to be the major effector mechanism in xenograft models. Here, using syngeneic immunocompetent mouse tumor models, we reveal that the therapeutic effects of CD47 blockade depend on dendritic cell but not macrophage cross-priming of T cell responses. The therapeutic effects of anti-CD47 antibody therapy were abrogated in T cell-deficient mice. In addition, the antitumor effects of CD47 blockade required expression of the cytosolic DNA sensor STING, but neither MyD88 nor TRIF, in CD11c(+) cells, suggesting that cytosolic sensing of DNA from tumor cells is enhanced by anti-CD47 treatment, further bridging the innate and adaptive responses. Notably, the timing of administration of standard chemotherapy markedly impacted the induction of antitumor T cell responses by CD47 blockade. Together, our findings indicate that CD47 blockade drives T cell-mediated elimination of immunogenic tumors.
in vivo CD47 blockade
Shi, L., et al. (2015). "CD47 deficiency ameliorates autoimmune nephritis in Fas(lpr) mice by suppressing IgG autoantibody production" J Pathol 237(3): 285-295. PubMed
CD47, a self-recognition marker, plays an important role in both innate and adaptive immune responses. To explore the potential role of CD47 in activation of autoreactive T and B cells and the production of autoantibodies in autoimmune disease, especially systemic lupus erythematosus (SLE), we have generated CD47 knockout Fas(lpr) (CD47(-/-) -Fas(lpr) ) mice and examined histopathological changes in the kidneys, cumulative survival rates, proteinuria, extent of splenomegaly and autoantibodies, serum chemistry and immunological parameters. In comparison with Fas(lpr) mice, CD47(-/-) -Fas(lpr) mice exhibit a prolonged lifespan and delayed autoimmune nephritis, including glomerular cell proliferation, basement membrane thickening, acute tubular atrophy and vacuolization. CD47(-/-) -Fas(lpr) mice have lower levels of proteinuria, associated with reduced deposition of complement C3 and C1q, and IgG but not IgM in the glomeruli, compared to age-matched Fas(lpr) mice. Serum levels of antinuclear antibodies and anti-double-stranded DNA antibodies are significantly lower in CD47(-/-) -Fas(lpr) than in Fas(lpr) mice. CD47(-/-) -Fas(lpr) mice also display less pronounced splenomegaly than Fas(lpr) mice. The mechanistic studies further suggest that CD47 deficiency impairs the antigenic challenge-induced production of IgG but not IgM, and that this effect is associated with reduction of T follicular cells and impairment of germinal centre development in lymphoid tissues. In conclusion, our results demonstrate that CD47 deficiency ameliorates lupus nephritis in Fas(lpr) mice via suppression of IgG autoantibody production. Copyright (c) 2015 Pathological Society of Great Britain and Ireland. Published by John Wiley & Sons, Ltd.
in vitro CD47 blockade, Immunofluorescence
Hsieh, C. P., et al. (2015). "Deficits in cerebellar granule cell development and social interactions in CD47 knockout mice" Dev Neurobiol 75(5): 463-484. PubMed
CD47 is involved in neurite differentiation in cultured neurons, but the function of CD47 in brain development is largely unknown. We determined that CD47 mRNA was robustly expressed in the developing cerebellum, especially in granule cells. CD47 protein was mainly expressed in the inner layer of the external granule layer (EGL), molecular layer, and internal granule layer (IGL), where granule cells individually become postmitotic and migrate, leading to neurite fasciculation. At postnatal day 8 (P8), CD47 knockout mice exhibited an increased number of proliferating granule cells in the EGL, whereas the CD47 agonist peptide 4N1K increased the number of postmitotic cells in primary granule cells. Knocking out the CD47 gene and anti-CD47 antibody impaired the radial migration of granule cells from the EGL to the IGL individually in mice and slice cultures. In primary granule cells, knocking out CD47 reduced the number of axonal collaterals and dendritic branches; by contrast, overexpressing CD47 or 4N1K treatment increased the axonal length and numbers of axonal collaterals and dendritic branches. Furthermore, the length of the fissure between Lobules VI and VII was decreased in CD47 knockout mice at P21 and at 14 wk after birth. Lastly, CD47 knockout mice exhibited increased social interaction at P21 and depressive-like behaviors at 10 wk after birth. Our study revealed that the cell adhesion molecule CD47 participates in multiple phases of granule cell development, including proliferation, migration, and neurite differentiation implying that aberrations of CD47 are risk factors that cause abnormalities in cerebellar development and atypical behaviors.
in vivo CD47 blockade
Maute, R. L., et al. (2015). "Engineering high-affinity PD-1 variants for optimized immunotherapy and immuno-PET imaging" Proc Natl Acad Sci U S A 112(47): E6506-6514. PubMed
Signaling through the immune checkpoint programmed cell death protein-1 (PD-1) enables tumor progression by dampening antitumor immune responses. Therapeutic blockade of the signaling axis between PD-1 and its ligand programmed cell death ligand-1 (PD-L1) with monoclonal antibodies has shown remarkable clinical success in the treatment of cancer. However, antibodies have inherent limitations that can curtail their efficacy in this setting, including poor tissue/tumor penetrance and detrimental Fc-effector functions that deplete immune cells. To determine if PD-1:PD-L1-directed immunotherapy could be improved with smaller, nonantibody therapeutics, we used directed evolution by yeast-surface display to engineer the PD-1 ectodomain as a high-affinity (110 pM) competitive antagonist of PD-L1. In contrast to anti-PD-L1 monoclonal antibodies, high-affinity PD-1 demonstrated superior tumor penetration without inducing depletion of peripheral effector T cells. Consistent with these advantages, in syngeneic CT26 tumor models, high-affinity PD-1 was effective in treating both small (50 mm(3)) and large tumors (150 mm(3)), whereas the activity of anti-PD-L1 antibodies was completely abrogated against large tumors. Furthermore, we found that high-affinity PD-1 could be radiolabeled and applied as a PET imaging tracer to efficiently distinguish between PD-L1-positive and PD-L1-negative tumors in living mice, providing an alternative to invasive biopsy and histological analysis. These results thus highlight the favorable pharmacology of small, nonantibody therapeutics for enhanced cancer immunotherapy and immune diagnostics.
in vitro CD47 blockade
Vermeer, D. W., et al. (2013). "Radiation-induced loss of cell surface CD47 enhances immune-mediated clearance of human papillomavirus-positive cancer" Int J Cancer 133(1): 120-129. PubMed
The increasing incidence of human papillomavirus (HPV) related oropharyngeal squamous cell carcinoma (OSSC) demands development of novel therapies. Despite presenting at a more advanced stage, HPV(+) oropharyngeal squamous cell carcinoma (OSCC) have a better prognosis than their HPV(-) counterparts. We have previously demonstrated that clearance of HPV(+) OSCC during treatment with radiation and chemotherapy requires an immune response which is likely responsible for the improved clinical outcomes. To further elucidate the mechanism of immune-mediated clearance, we asked whether radiation therapy induces tumor cell changes that allow the body to recognize and aid in tumor clearance. Here, we describe a radiation-induced change in tumor surface protein expression that is critical for immune-mediated clearance. Radiation therapy decreases surface expression of CD47, a self-marker. CD47 is frequently overexpressed in head and neck squamous cell carcinoma and radiation induces a decrease of CD47 in a dose-dependent manner. We show that both in vitro and in vivo tumor cell CD47 protein levels are restored over time after sublethal radiation exposure and that protein levels on adjacent, normal tissues remain unaffected. Furthermore, reduction of tumor cell CD47 increases phagocytosis of these cells by dendritic cells and leads to increased interferon gamma and granzyme production from mixed lymphocytes. Finally, decreasing tumor cell CD47 in combination with standard radiation and chemotherapy results in improved immune-mediated tumor clearance in vivo. These findings help define an important mechanism of radiation-related immune clearance and suggest that decreasing CD47 specifically on tumor cells may be a good therapeutic target for HPV related disease.
in vitro CD47 blockade
Majeti, R., et al. (2009). "CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells" Cell 138(2): 286-299. PubMed
Acute myeloid leukemia (AML) is organized as a cellular hierarchy initiated and maintained by a subset of self-renewing leukemia stem cells (LSC). We hypothesized that increased CD47 expression on human AML LSC contributes to pathogenesis by inhibiting their phagocytosis through the interaction of CD47 with an inhibitory receptor on phagocytes. We found that CD47 was more highly expressed on AML LSC than their normal counterparts, and that increased CD47 expression predicted worse overall survival in three independent cohorts of adult AML patients. Furthermore, blocking monoclonal antibodies directed against CD47 preferentially enabled phagocytosis of AML LSC and inhibited their engraftment in vivo. Finally, treatment of human AML LSC-engrafted mice with anti-CD47 antibody depleted AML and targeted AML LSC. In summary, increased CD47 expression is an independent, poor prognostic factor that can be targeted on human AML stem cells with blocking monoclonal antibodies capable of enabling phagocytosis of LSC.
in vivo CD47 blockade
Chang, H. P., et al. (2001). "Functional blocking of integrin-associated protein impairs memory retention and decreases glutamate release from the hippocampus" Neuroscience 102(2): 289-296. PubMed
We have previously demonstrated that integrin-associated protein is involved in memory consolidation of one-way inhibitory avoidance learning in rats and mice. In the present study, we examined the effects of functional blocking of integrin-associated protein on memory retention, long-term potentiation and glutamate release in mice as well as on cell attachment to extracellular matrix protein in primary cultures. The results indicated that integrin-associated protein monoclonal antibody miap301, when directly injected into the dentate gyrus of the hippocampus at moderate doses, significantly impairs memory retention in mice in the same one-way inhibitory avoidance task and decreases the amplitude of tetanic stimulation-induced long-term potentiation in dentate gyrus neurons. At a dose that effectively impairs both memory retention and long-term potentiation, integrin-associated protein monoclonal antibody also significantly blocks potassium chloride-induced glutamate release from the hippocampus in vivo. Results from western blot confirmed the presence of integrin-associated protein at the synaptic area. Cell adhesion experiments further revealed that integrin-associated protein monoclonal antibody markedly inhibits granular cell attachment to thrombospondin, the extracellular matrix protein known to bind integrin-associated protein, but not to collagen and laminin, the extracellular matrix proteins known to bind integrin. From these results we suggest that integrin-associated protein monoclonal antibody may impair synaptic plasticity and behavioral plasticity in mice through blockade of granular cell attachment to extracellular matrix protein and the subsequent signal transduction, and through inhibition of glutamate release from the hippocampus.