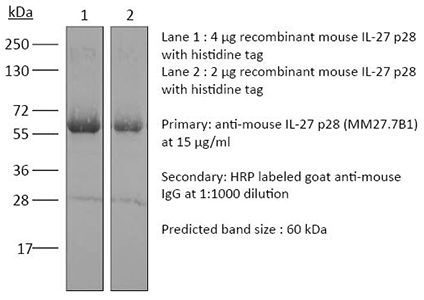
InVivoMAb anti-mouse IL-27 p28
Product Details
The MM27.7B1 monoclonal antibody reacts with the p28 subunit of mouse IL-27, a member of the IL-6/IL-12 cytokine family. IL-27 is a heterodimeric cytokine composed the EBI3 (Epstein-Barr virus induced gene 3) protein and the p28 subunit, also known as IL-30. Myeloid cells including macrophages, inflammatory monocytes, microglia, and dendritic cells are the dominant cellular sources of IL-27. These cells secrete IL-27 in response to TLR ligands and inflammatory cytokines. IL-27 p28 signals through IL-27 receptors (IL-27R) which are highly expressed by NK cells and activated T cells and to a lesser extent on B cells, naïve T cells, and others. IL-27 signaling has been shown to have both pro-inflammatory and anti-inflammatory effects. Signaling through IL-27R promotes Th1 polarization and IFNγ production but also suppresses the differentiation and proliferation of Th2 and Th17 cells and induces the expression of IL-10. The MM27.7B1 clone has been shown to neutralize the bioactivity of mouse IL-27 in vivo and in vitro and reported to also bind and inhibit human IL-27.Specifications
| Isotype | Mouse IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG2a isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse IL-27 |
| Reported Applications |
in vivo IL-27 p28 neutralization in vitro IL-27 p28 neutralization Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein A |
| RRID | AB_2819053 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vivo IL-27 p28 neutralization
Ayasoufi, K., et al. (2019). "Interleukin-27 promotes CD8+ T cell reconstitution following antibody-mediated lymphoablation" JCI Insight 4(7). PubMed
Antibody-mediated lymphoablation is used in solid organ and stem cell transplantation and autoimmunity. Using murine anti-thymocyte globulin (mATG) in a mouse model of heart transplantation, we previously reported that the homeostatic recovery of CD8+ T cells requires help from depletion-resistant memory CD4+ T cells delivered through CD40-expressing B cells. This study investigated the mechanisms by which B cells mediate CD8+ T cell proliferation in lymphopenic hosts. While CD8+ T cell recovery required MHC class I expression in the host, the reconstitution occurred independently of MHC class I, MHC class II, or CD80/CD86 expression on B cells. mATG lymphoablation upregulated the B cell expression of several cytokine genes, including IL-15 and IL-27, in a CD4-dependent manner. Neither treatment with anti-CD122 mAb nor the use of IL-15Ralpha-/- recipients altered CD8+ T cell recovery after mATG treatment, indicating that IL-15 may be dispensable for T cell proliferation in our model. Instead, IL-27 neutralization or the use of IL-27Ralpha-/- CD8+ T cells inhibited CD8+ T cell proliferation and altered the phenotype and cytokine profile of reconstituted CD8+ T cells. Our findings uncover what we believe is a novel role of IL-27 in lymphopenia-induced CD8+ T cell proliferation and suggest that targeting B cell-derived cytokines may increase the efficacy of lymphoablation and improve transplant outcomes.
in vivo IL-27 p28 neutralization
Xin, G., et al. (2018). "Single-cell RNA sequencing unveils an IL-10-producing helper subset that sustains humoral immunity during persistent infection" Nat Commun 9(1): 5037. PubMed
During chronic viral infection, the inflammatory function of CD4 T-cells becomes gradually attenuated. Concurrently, Th1 cells progressively acquire the capacity to secrete the cytokine IL-10, a potent suppressor of antiviral T cell responses. To determine the transcriptional changes that underlie this adaption process, we applied a single-cell RNA-sequencing approach and assessed the heterogeneity of IL-10-expressing CD4 T-cells during chronic infection. Here we show an IL-10-producing population with a robust Tfh-signature. Using IL-10 and IL-21 double-reporter mice, we further demonstrate that IL-10(+)IL-21(+)co-producing Tfh cells arise predominantly during chronic but not acute LCMV infection. Importantly, depletion of IL-10(+)IL-21(+)co-producing CD4 T-cells or deletion of Il10 specifically in Tfh cells results in impaired humoral immunity and viral control. Mechanistically, B cell-intrinsic IL-10 signaling is required for sustaining germinal center reactions. Thus, our findings elucidate a critical role for Tfh-derived IL-10 in promoting humoral immunity during persistent viral infection.
in vivo IL-27 p28 neutralization, Flow Cytometry
Clement, M., et al. (2016). "Cytomegalovirus-Specific IL-10-Producing CD4+ T Cells Are Governed by Type-I IFN-Induced IL-27 and Promote Virus Persistence" PLoS Pathog 12(12): e1006050. PubMed
CD4+ T cells support host defence against herpesviruses and other viral pathogens. We identified that CD4+ T cells from systemic and mucosal tissues of hosts infected with the beta-herpesviridae human cytomegalovirus (HCMV) or murine cytomegalovirus (MCMV) express the regulatory cytokine interleukin (IL)-10. IL-10+CD4+ T cells co-expressed TH1-associated transcription factors and chemokine receptors. Mice lacking T cell-derived IL-10 elicited enhanced antiviral T cell responses and restricted MCMV persistence in salivary glands and secretion in saliva. Thus, IL-10+CD4+ T cells suppress antiviral immune responses against CMV. Expansion of this T-cell population in the periphery was promoted by IL-27 whereas mucosal IL-10+ T cell responses were ICOS-dependent. Infected Il27ralpha-deficient mice with reduced peripheral IL-10+CD4+ T cell accumulation displayed robust T cell responses and restricted MCMV persistence and shedding. Temporal inhibition experiments revealed that IL-27R signaling during initial infection was required for the suppression of T cell immunity and control of virus shedding during MCMV persistence. IL-27 production was promoted by type-I IFN, suggesting that beta-herpesviridae exploit the immune-regulatory properties of this antiviral pathway to establish chronicity. Further, our data reveal that cytokine signaling events during initial infection profoundly influence virus chronicity.
in vivo IL-27 p28 neutralization
Anderson, A. C., et al. (2015). "A T cell extrinsic mechanism by which IL-2 dampens Th17 differentiation" J Autoimmun 59: 38-42. PubMed
Genetic variants in il2 and il2ra have been associated with autoimmune disease susceptibility in both genome-wide association studies (GWAS) in humans and in genetic linkage studies in experimental models of autoimmunity. Specifically, genetic variants resulting in a low IL-2 phenotype are susceptibility alleles while variants resulting in a high IL-2 phenotype are resistance alleles. The association of high IL-2 phenotypes with resistance has been attributed primarily to the T cell intrinsic promotion of regulatory T cell development, maintenance, and function; however, IL-2 can also act T cell intrinsically to dampen differentiation of pathogenic IL-17-producing Th17 cells. Here, we have uncovered a novel T cell extrinsic mechanism whereby IL-2 promotes both IFN-gamma and IL-27 production from tissue resident macrophages which in turn dampen the differentiation of pathogenic Th17 cells.
in vivo IL-27 p28 neutralization
Marillier, R. G., et al. (2014). "IL-27p28 is essential for parent-to-F1 acute graft-versus-host disease" Eur J Immunol 44(7): 2064-2073. PubMed
Acute graft versus host disease (aGVHD) remains a life-threatening complication of bone marrow transplantation. Here we show that IL-27, a member of the IL-12 cytokine family, plays an essential role in a parent-to-F1 murine aGVHD model, using B6 mice as parents and B6D2 mice as F1 recipients. IL-27 is transiently detectable in the serum of B6D2 recipients of B6 spleen cells, with a peak at day 10. Treatment with anti-IL-27p28 mAb MM27.7B1 (alphap28Ab), at the time of and six days after B6 cell transfer, blocked GVHD. Protection was associated with host cell survival and undiminished engraftment of donor cells, lack of host B-cell depletion, increased Th2-type immunoglobulin production, a decrease in serum IFN-gamma, a drop in anti-H-2D(d) cytotoxic T lymphocyte activity and an increase in Foxp3(+) T cells. We therefore conclude that IL-27 plays a critical role in the parent-to-F1 model of aGVHD and that blocking IL-27 could have therapeutic relevance.
Flow Cytometry
Bosmann, M., et al. (2014). "Tyrosine kinase 2 promotes sepsis-associated lethality by facilitating production of interleukin-27" J Leukoc Biol 96(1): 123-131. PubMed
The aim of this study was to test the hypothesis that gene expression and release of IL-27 may be modulated by Tyk2. Macrophages derived from the peritoneum or bone marrow of C57BL/10SnJ (WT) mice produced abundant amounts of IL-27(p28) following TLR4 activation by LPS. In contrast, production of IL-27(p28), but not EBI3, was reduced by approximately 50% in TLR4-activated macrophages derived from mice with genetic deficiency of Tyk2 compared with WT macrophages. Frequencies of IL-27(p28)+F4/80+CD11b+ cells were lower in TLR4-activated macrophages derived from Tyk2-/- mice. Mechanistically, Tyk2-/- resulted in disruption of a type I IFN-dependent mechanism for production of IL-27(p28), which was induced by type I IFNs, and release of IL-27 was defective in macrophages from IFN-beta-/- and IFNAR1-/- mice. In contrast, Tyk2 was not required to mediate the effects of IL-27 on target gene expression in CD4(+) T cells. In vivo, we observed that Tyk2-/- mice have improved survival following endotoxic shock or polymicrobial sepsis induced by CLP. Plasma levels of IL-27(p28) during endotoxic shock or polymicrobial sepsis were markedly reduced in Tyk2-/- mice compared with WT mice. Disruption of IL-27 signaling using IL-27RA-/- mice was protective against sepsis-associated mortality. These data suggest that Tyk2 may mediate adverse outcomes of SIRS by promoting the production of IL-27. In conclusion, this report identifies Tyk2 as a prerequisite factor in the molecular networks that are involved in generation of IL-27.