InVivoMAb anti-human PD-1 (CD279)
Product Details
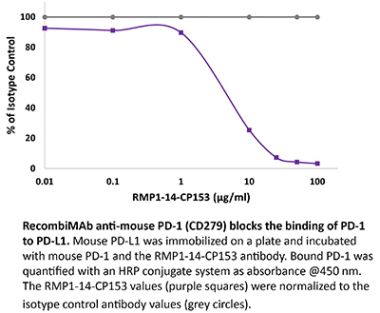
The J110 monoclonal antibody reacts with human PD-1 (programmed death-1) also known as CD279. PD-1 is a 50-55 kDa cell surface receptor encoded by the Pdcd1 gene that belongs to the CD28 family of the Ig superfamily. PD-1 is transiently expressed on CD4 and CD8 thymocytes as well as activated T and B lymphocytes and myeloid cells. PD-1 expression declines after successful elimination of antigen. Additionally, Pdcd1 mRNA is expressed in developing B lymphocytes during the pro-B-cell stage. PD-1’s structure includes a ITIM (immunoreceptor tyrosine-based inhibitory motif) suggesting that PD-1 negatively regulates TCR signals. PD-1 signals via binding its two ligands, PD-L1 and PD-L2 both members of the B7 family. Upon ligand binding, PD-1 signaling inhibits T-cell activation, leading to reduced proliferation, cytokine production, and T cell death. Additionally, PD-1 is known to play key roles in peripheral tolerance and prevention of autoimmune disease in mice as PD-1 knockout animals show dilated cardiomyopathy, splenomegaly, and loss of peripheral tolerance. Induced PD-L1 expression is common in many tumors including squamous cell carcinoma, colon adenocarcinoma, and breast adenocarcinoma. PD-L1 overexpression results in increased resistance of tumor cells to CD8 T cell mediated lysis. In mouse models of melanoma, tumor growth can be transiently arrested via treatment with antibodies which block the interaction between PD-L1 and its receptor PD-1. For these reasons anti-PD-1 mediated immunotherapies are currently being explored as cancer treatments.Specifications
| Isotype | Mouse IgG1, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG1 isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Not available or unknown |
| Reported Applications |
in vivo PD-1 blockade in humanized mice Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_10950168 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vivo PD-1 blockade in humanized mice
Sanlorenzo, M., et al. (2018). "BRAF and MEK Inhibitors Increase PD-1-Positive Melanoma Cells Leading to a Potential Lymphocyte-Independent Synergism with Anti-PD-1 Antibody" Clin Cancer Res 24(14): 3377-3385. PubMed
Purpose: BRAF and MEK inhibitors (BRAF/MEKi) favor melanoma-infiltrating lymphocytes, providing the rationale for current combinatorial trials with anti-PD-1 antibody. A portion of melanoma cells may express PD-1, and anti-PD-1 antibody could have a direct antitumor effect. Here, we explore whether BRAF/MEKi modulate rates of PD-1(+) melanoma cells, supporting an additional-lymphocyte-independent-basis for their therapeutic combination with anti-PD-1 antibody.Experimental Design: With data mining and flow cytometry, we assessed PD-1, PD-L1/2 expression on melanoma cell lines (CCLE, N = 61; validation cell lines, N = 7) and melanoma tumors (TCGA, N = 214). We explored in vitro how BRAF/MEKi affect rates of PD-1(+), PD-L1/2(+) melanoma cells, and characterized the proliferative and putative stemness features of PD-1(+) melanoma cells. We tested the functional lymphocyte-independent effect of anti-PD-1 antibody alone and in combination with BRAF/MEKi in vitro and in an in vivo immunodeficient murine model.Results: PD-1 is consistently expressed on a small subset of melanoma cells, but PD-1(+) cells increase to relevant rates during BRAF/MEKi treatment [7.3% (5.6-14.2) vs. 1.5% (0.7-3.2), P = 0.0156; N = 7], together with PD-L2(+) melanoma cells [8.5% (0.0-63.0) vs. 1.5% (0.2-43.3), P = 0.0312; N = 7]. PD-1(+) cells proliferate less than PD-1(-) cells (avg. 65% less; t = 7 days) and are preferentially endowed with stemness features. In vivo, the direct anti-melanoma activity of PD-1 blockage as monotherapy was negligible, but its association with BRAF/MEKi significantly delayed the development of drug resistance and tumor relapse.Conclusions: BRAF/MEKi increase the rates of PD-1(+) melanoma cells that may sustain tumor relapse, providing a lymphocyte-independent rationale to explore combinatory strategies with anti-PD-1 antibody. Clin Cancer Res; 24(14); 3377-85.
Flow Cytometry
Yamane, H., et al. (2015). "Programmed cell death protein 1 and programmed death-ligand 1 are expressed on the surface of some small-cell lung cancer lines" Am J Cancer Res 5(4): 1553-1557. PubMed
INTRODUCTION: Programmed cell death protein 1 (PD-1) and programmed death-ligand 1 (PD-L1) play a major role in suppressing the immune system during the formation of the PD-1/PD-L1 pathway, which transmits an inhibitory signal to reduce T cell activity. PD-L1 is often expressed in various malignant tumors. In contrast, PD-1 is generally observed in activated lymphocytes and myeloid-derived dendritic cells. Of the malignant cells, only Jurkat cells under special conditions and angioimmunoblastic T-cell lymphoma tissue cells express PD-1 on their surface. METHODS: To clarify whether the PD-1/PD-L1 pathway participates in the immunotolerance of small-cell lung cancer (SCLC) cells, we examined the expressions of PD-1 and PD-L1 on the cell surface of SCLC cell lines using flow cytometry and reverse transcription polymerase chain reaction. RESULTS: Among the four SCLC cell lines examined, only SBC-3 expressed both PD-1 and PD-L1. CONCLUSIONS: We demonstrated that both PD-1 and PD-L1 molecules were co-expressed on the surface of SCLC cells. Although the biological implications of this remain unclear, we speculate that PD-1 and its ligand on the SCLC cells may participate in the growth inhibition of tumor cells as reported in cytotoxic T cells.
Flow Cytometry
Bennett, F., et al. (2003). "Program death-1 engagement upon TCR activation has distinct effects on costimulation and cytokine-driven proliferation: attenuation of ICOS, IL-4, and IL-21, but not CD28, IL-7, and IL-15 responses" J Immunol 170(2): 711-718. PubMed
The program death 1 (PD-1) receptor and its ligands, PD-1 ligand (PD-L)1 and PD-L2, define a novel regulatory pathway with potential inhibitory effects on T, B, and monocyte responses. In the present study, we show that human CD4(+) T cells express PD-1, PD-L1, and PD-L2 upon activation, and Abs to the receptor can be agonists or antagonists of the pathway. Under optimal conditions of stimulation, ICOS but not CD28 costimulation can be prevented by PD-1 engagement. IL-2 levels induced by costimulation are critical in determining the outcome of the PD-1 engagement. Thus, low to marginal IL-2 levels produced upon ICOS costimulation account for the greater sensitivity of this pathway to PD-1-mediated inhibition. Interestingly, exogenous IL-2, IL-7, and IL-15 but not IL-4 and IL-21 can rescue PD-1 inhibition, suggesting that among these cytokines only those that activate STAT5 can rescue PD-1 inhibition. As STAT5 has been implicated in the maintenance of IL-2Ralpha expression, these results suggest that IL-7 and IL-15 restore proliferation under conditions of PD-1 engagement by enhancing high-affinity IL-2R expression and hence, IL-2 responsiveness.