InVivoMAb anti-mouse CD71 (TfR1)
Product Details
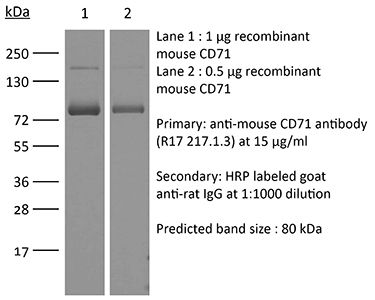
The R17 217.1.3/TIB-219 monoclonal antibody reacts with mouse CD71 also known as transferrin receptor protein 1 (TfR1). CD71 is a 170-180 kDa type II homodimeric transmembrane glycoprotein which is expressed on the surface of proliferating cells, reticulocytes, and erythroid precursors. CD71 plays a role in the control of cellular proliferation and is required for iron import from transferrin into cells by endocytosis. Due to its important role in proliferation and cellular iron intake as well as the fact that many cancerous cells express high levels of CD71 it is being explored as a potential new target in cases of human leukemia & lymphoma. The R17 217.1.3/TIB-219 antibody has been shown to deplete CD71+ erythroid splenocytes.Specifications
| Isotype | Rat IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG2a isotype control, anti-trinitrophenol |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse erythroleukemia cell line 745.6 |
| Reported Applications | in vivo depletion of CD71+ cells |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_10950526 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Additional Formats
Recommended Products
in vivo depletion of CD71+ cells
Torow, N., et al. (2015). "Active suppression of intestinal CD4(+)TCRalphabeta(+) T-lymphocyte maturation during the postnatal period" Nat Commun 6: 7725. PubMed
Priming of the mucosal immune system during the postnatal period substantially influences host-microbial interaction and susceptibility to immune-mediated diseases in adult life. The underlying mechanisms are ill defined. Here we show that shortly after birth, CD4 T cells populate preformed lymphoid structures in the small intestine and quickly acquire a distinct transcriptional profile. T-cell recruitment is independent of microbial colonization and innate or adaptive immune stimulation but requires beta7 integrin expression. Surprisingly, neonatal CD4 T cells remain immature throughout the postnatal period under homeostatic conditions but undergo maturation and gain effector function on barrier disruption. Maternal SIgA and regulatory T cells act in concert to prevent immune stimulation and maintain the immature phenotype of CD4 T cells in the postnatal intestine during homeostasis. Active suppression of CD4 T-cell maturation during the postnatal period might contribute to prevent auto-reactivity, sustain a broad TCR repertoire and establish life-long immune homeostasis.
in vivo depletion of CD71+ cells
Wynn, J. L., et al. (2015). "Neonatal CD71+ Erythroid Cells Do Not Modify Murine Sepsis Mortality" J Immunol 195(3): 1064-1070. PubMed
Sepsis is a major cause of neonatal mortality and morbidity worldwide. A recent report suggested that murine neonatal host defense against infection could be compromised by immunosuppressive CD71(+) erythroid splenocytes. We examined the impact of CD71(+) erythroid splenocytes on murine neonatal mortality to endotoxin challenge or polymicrobial sepsis and characterized circulating CD71(+) erythroid (CD235a(+)) cells in human neonates. Adoptive transfer or an Ab-mediated reduction in neonatal CD71(+) erythroid splenocytes did not alter murine neonatal survival to endotoxin challenge or polymicrobial sepsis challenge. Ex vivo immunosuppression of stimulated adult CD11b(+) cells was not limited to neonatal splenocytes; it also occurred with adult and neonatal bone marrow. Animals treated with anti-CD71 Ab showed reduced splenic bacterial load following bacterial challenge compared with isotype-treated mice. However, adoptive transfer of enriched CD71(+) erythroid splenocytes to CD71(+)-reduced animals did not reduce bacterial clearance. Human CD71(+)CD235a(+) cells were common among cord blood mononuclear cells and were shown to be reticulocytes. In summary, a lack of effect on murine survival to polymicrobial sepsis following adoptive transfer or diminution of CD71(+) erythroid splenocytes under these experimental conditions suggests that the impact of these cells on neonatal infection risk and progression may be limited. An unanticipated immune priming effect of anti-CD71 Ab treatment, rather than a reduction in immunosuppressive CD71(+) erythroid splenocytes, was likely responsible for the reported enhanced bacterial clearance. In humans, the well-described rapid decrease in circulating reticulocytes after birth suggests that they may have a limited role in reducing inflammation secondary to microbial colonization.