InVivoPlus anti-mouse TIM-3 (CD366)
Product Details
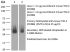
The RMT3-23 monoclonal antibody reacts with mouse TIM-3 (T cell immunoglobulin and mucin domain-3) also known as CD366. TIM-3 is a 60 kDa member of the TIM family of immune checkpoint receptors and exists as a type I transmembrane glycoprotein with a mucin-like domain in its extracellular portion and a tyrosine phosphorylation motif in its cytoplasmic portion. TIM-3 is specifically expressed at high levels on the surface of Th1 lymphocytes whereas Th2 lymphocytes express TIM-1 and TIM-2. TIM-3 activation occurs via binding to the cell-associated C-type lectin galectin-9. Upon binding TIM-3 induces apoptosis of Th1 cells. Inhibition of TIM-3 signaling in mice has been shown to exacerbate experimental autoimmune encephalomyelitis, promote IFNγ production and Th1 cell proliferation. Tim-3 has also been shown to be required for the induction of tolerance, as both TIM-3 knockout animals and mice treated with TIM-3-Ig fusion protein display defects in the induction of antigen-specific tolerance. Additionally, TIM-3 signaling is currently being explored as a cancer immunotherapy target as CD8 T cells which express both TIM-3 and PD-1 exhibit greater defects in both cell-cycle progression and effector cytokine production than cells that express PD-1 alone. The RMT3-23 antibody acts as a TIM-3 receptor antagonist and has been shown to have functional activity including suppressing tumor cell growth in a murine sarcoma model.Specifications
| Isotype | Rat IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoPlus rat IgG2a isotype control, anti-trinitrophenol |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Recombinant mouse TIM-3 |
| Reported Applications |
in vivo TIM-3 neutralization in vitro TIM-3 blocking Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin* |
<1EU/mg (<0.001EU/μg) Determined by LAL gel clotting assay |
| Aggregation* |
<5% Determined by SEC |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_10949464 |
| Molecular Weight | 150 kDa |
| Murine Pathogen Tests* |
Ectromelia/Mousepox Virus: Negative Hantavirus: Negative K Virus: Negative Lactate Dehydrogenase-Elevating Virus: Negative Lymphocytic Choriomeningitis virus: Negative Mouse Adenovirus: Negative Mouse Cytomegalovirus: Negative Mouse Hepatitis Virus: Negative Mouse Minute Virus: Negative Mouse Norovirus: Negative Mouse Parvovirus: Negative Mouse Rotavirus: Negative Mycoplasma Pulmonis: Negative Pneumonia Virus of Mice: Negative Polyoma Virus: Negative Reovirus Screen: Negative Sendai Virus: Negative Theiler’s Murine Encephalomyelitis: Negative |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Additional Formats
Recommended Products
in vivo TIM-3 neutralization
Liu, J. F., et al. (2018). "Blockade of TIM3 relieves immunosuppression through reducing regulatory T cells in head and neck cancer" J Exp Clin Cancer Res 37(1): 44. PubMed
BACKGROUND: T-cell immunoglobulin mucin 3 (TIM3) is a negative immune checkpoint and plays a crucial part in tumor-induced immune suppression. However, the mechanism of TIM3 in regulating immunosuppression in head and neck squamous cell carcinoma (HNSCC) was still not quite clear. METHODS: We carried out the immunohistochemistry staining of HNSCC tissue microarrays. Through quantification of the histoscore, we performed the correlation analysis among the TIM3, Galectin-9, Foxp3, CD68 and CD163. The effects of TIM3 on regulatory T cells (Tregs) and macrophages were detected by utilizing the Tgfbr1/Pten 2cKO HNSCC mouse model. Flow cytometry were used to analysis the percent of Tregs, macrophages and IFN-gamma. RESULTS: We demonstrated the close association among TIM3/Galectin-9 pathway, regulatory T cell marker (Foxp3) and macrophage marker (CD68, CD163) in human HNSCC. In the transgenic HNSCC mouse model, blockade of TIM3 by the anti-TIM3 monoclonal antibody induced a reduction of CD4(+)CD25(+)Foxp3(+) Tregs. Meanwhile, the population of TIM3(+) Tregs was also decreased. However, the population of CD206(+) macrophages was not significantly declined. The increased IFN-gamma production on CD8(+) T cells in anti-TIM3 treatment mice showed that the antitumor immune response was enhanced through suppression of these negative immune factors. CONCLUSIONS: The present study demonstrated that TIM3 was associated with the immunosuppression in HNSCC. And targeting TIM3 can enhance anti-tumor immune response by decreasing Tregs in HNSCC.
in vivo TIM-3 neutralization
Ngiow, S. F., et al. (2015). "A Threshold Level of Intratumor CD8+ T-cell PD1 Expression Dictates Therapeutic Response to Anti-PD1" Cancer Res 75(18): 3800-3811. PubMed
Despite successes, thus far, a significant proportion of the patients treated with anti-PD1 antibodies have failed to respond. We use mouse tumor models of anti-PD1 sensitivity and resistance and flow cytometry to assess tumor-infiltrating immune cells immediately after therapy. We demonstrate that the expression levels of T-cell PD1 (PD1(lo)), myeloid, and T-cell PDL1 (PDL1(hi)) in the tumor microenvironment inversely correlate and dictate the efficacy of anti-PD1 mAb and function of intratumor CD8(+) T cells. In sensitive tumors, we reveal a threshold for PD1 downregulation on tumor-infiltrating CD8(+) T cells below which the release of adaptive immune resistance is achieved. In contrast, PD1(hi) T cells in resistant tumors fail to be rescued by anti-PD1 therapy and remain dysfunctional unless intratumor PDL1(lo) immune cells are targeted. Intratumor Tregs are partly responsible for the development of anti-PD1-resistant tumors and PD1(hi) CD8(+) T cells. Our analyses provide a framework to interrogate intratumor CD8(+) T-cell PD1 and immune PDL1 levels and response in human cancer. Cancer Res; 75(18); 3800-11. (c)2015 AACR.
in vivo TIM-3 neutralization
Tripathi, S., et al. (2015). "Effect of TIM-3 Blockade on the Immunophenotype and Cytokine Profile of Murine Uterine NK Cells" PLoS One 10(4): e0123439. PubMed
NK cells are the most abundant lymphocyte population in the feto-maternal interface during gestation. The uterine NK cells (uNK) are transient, have a unique immunophenotype and produce a number of cytokines. These cytokines play an important role in establishment and maintenance of vascular remodeling and tolerance associated with successful pregnancy. The uNK cells also express TIM-3 during gestation and blockade of TIM-3 expression results in fetal loss in mice. In this study we determined the effect of TIM-3 blockade on uNK cells. Specifically we observed surface receptor phenotype and cytokine production by uNK cells following TIM-3 blockade. Our results show that TIM-3 plays a role in regulating the uNK cells and contributes to the maintenance of tolerance at the feto-maternal interface.
in vivo TIM-3 neutralization
Kurtulus, S., et al. (2015). "TIGIT predominantly regulates the immune response via regulatory T cells" J Clin Invest. doi : 10.1172/JCI81187. PubMed
Coinhibitory receptors are critical for the maintenance of immune homeostasis. Upregulation of these receptors on effector T cells terminates T cell responses, while their expression on Tregs promotes their suppressor function. Understanding the function of coinhibitory receptors in effector T cells and Tregs is crucial, as therapies that target coinhibitory receptors are currently at the forefront of treatment strategies for cancer and other chronic diseases. T cell Ig and ITIM domain (TIGIT) is a recently identified coinhibitory receptor that is found on the surface of a variety of lymphoid cells, and its role in immune regulation is just beginning to be elucidated. We examined TIGIT-mediated immune regulation in different murine cancer models and determined that TIGIT marks the most dysfunctional subset of CD8+ T cells in tumor tissue as well as tumor-tissue Tregs with a highly active and suppressive phenotype. We demonstrated that TIGIT signaling in Tregs directs their phenotype and that TIGIT primarily suppresses antitumor immunity via Tregs and not CD8+ T cells. Moreover, TIGIT+ Tregs upregulated expression of the coinhibitory receptor TIM-3 in tumor tissue, and TIM-3 and TIGIT synergized to suppress antitumor immune responses. Our findings provide mechanistic insight into how TIGIT regulates immune responses in chronic disease settings.
in vivo TIM-3 neutralization
Mittal, D., et al. (2014). "Antimetastatic effects of blocking PD-1 and the adenosine A2A receptor" Cancer Res 74(14): 3652-3658. PubMed
Adenosine targeting is an attractive new approach to cancer treatment, but no clinical study has yet examined adenosine inhibition in oncology despite the safe clinical profile of adenosine A2A receptor inhibitors (A2ARi) in Parkinson disease. Metastasis is the main cause of cancer-related deaths worldwide, and therefore we have studied experimental and spontaneous mouse models of melanoma and breast cancer metastasis to demonstrate the efficacy and mechanism of a combination of A2ARi in combination with anti-PD-1 monoclonal antibody (mAb). This combination significantly reduces metastatic burden and prolongs the life of mice compared with either monotherapy alone. Importantly, the combination was only effective when the tumor expressed high levels of CD73, suggesting a tumor biomarker that at a minimum could be used to stratify patients that might receive this combination. The mechanism of the combination therapy was critically dependent on NK cells and IFNgamma, and to a lesser extent, CD8(+) T cells and the effector molecule, perforin. Overall, these results provide a strong rationale to use A2ARi with anti-PD-1 mAb for the treatment of minimal residual and metastatic disease.
in vivo TIM-3 neutralization
McGray, A. J., et al. (2014). "Immunotherapy-induced CD8+ T cells instigate immune suppression in the tumor" Mol Ther 22(1): 206-218. PubMed
Despite clear evidence of immunogenicity, cancer vaccines only provide a modest clinical benefit. To evaluate the mechanisms that limit tumor regression following vaccination, we have investigated the weak efficacy of a highly immunogenic experimental vaccine using a murine melanoma model. We discovered that the tumor adapts rapidly to the immune attack instigated by tumor-specific CD8+ T cells in the first few days following vaccination, resulting in the upregulation of a complex set of biological networks, including multiple immunosuppressive processes. This rapid adaptation acts to prevent sustained local immune attack, despite continued infiltration by increasing numbers of tumor-specific T cells. Combining vaccination with adoptive transfer of tumor-specific T cells produced complete regression of the treated tumors but did not prevent the adaptive immunosuppression. In fact, the adaptive immunosuppressive pathways were more highly induced in regressing tumors, commensurate with the enhanced level of immune attack. Examination of tumor infiltrating T-cell functionality revealed that the adaptive immunosuppression leads to a progressive loss in T-cell function, even in tumors that are regressing. These novel observations that T cells produced by therapeutic intervention can instigate a rapid adaptive immunosuppressive response within the tumor have important implications for clinical implementation of immunotherapies.
in vivo TIM-3 neutralization
Dolina, J. S., et al. (2014). "Liver-primed CD8+ T cells suppress antiviral adaptive immunity through galectin-9-independent T-cell immunoglobulin and mucin 3 engagement of high-mobility group box 1 in mice" Hepatology 59(4): 1351-1365. PubMed
The liver is a tolerogenic environment exploited by persistent infections, such as hepatitis B (HBV) and C (HCV) viruses. In a murine model of intravenous hepatotropic adenovirus infection, liver-primed antiviral CD8(+) T cells fail to produce proinflammatory cytokines and do not display cytolytic activity characteristic of effector CD8(+) T cells generated by infection at an extrahepatic, that is, subcutaneous, site. Importantly, liver-generated CD8(+) T cells also appear to have a T-regulatory (Treg) cell function exemplified by their ability to limit proliferation of antigen-specific T-effector (Teff ) cells in vitro and in vivo via T-cell immunoglobulin and mucin 3 (Tim-3) expressed by the CD8(+) Treg cells. Regulatory activity did not require recognition of the canonical Tim-3 ligand, galectin-9, but was dependent on CD8(+) Treg cell-surface Tim-3 binding to the alarmin, high-mobility group box 1 (HMGB-1). CONCLUSION: Virus-specific Tim-3(+) CD8(+) T cells operating through HMGB-1 recognition in the setting of acute and chronic viral infections of the liver may act to dampen hepatic T-cell responses in the liver microenvironment and, as a consequence, limit immune-mediated tissue injury or promote the establishment of persistent infections.
in vitro TIM-3 blocking, Flow Cytometry
Erickson, J. J., et al. (2014). "Programmed death-1 impairs secondary effector lung CD8(+) T cells during respiratory virus reinfection" J Immunol 193(10): 5108-5117. PubMed
Reinfections with respiratory viruses are common and cause significant clinical illness, yet precise mechanisms governing this susceptibility are ill defined. Lung Ag-specific CD8(+) T cells (T(CD8)) are impaired during acute viral lower respiratory infection by the inhibitory receptor programmed death-1 (PD-1). To determine whether PD-1 contributes to recurrent infection, we first established a model of reinfection by challenging B cell-deficient mice with human metapneumovirus (HMPV) several weeks after primary infection, and found that HMPV replicated to high titers in the lungs. A robust secondary effector lung TCD8 response was generated during reinfection, but these cells were more impaired and more highly expressed the inhibitory receptors PD-1, LAG-3, and 2B4 than primary T(CD8). In vitro blockade demonstrated that PD-1 was the dominant inhibitory receptor early after reinfection. In vivo therapeutic PD-1 blockade during HMPV reinfection restored lung T(CD8) effector functions (i.e., degranulation and cytokine production) and enhanced viral clearance. PD-1 also limited the protective efficacy of HMPV epitope-specific peptide vaccination and impaired lung T(CD8) during heterotypic influenza virus challenge infection. Our results indicate that PD-1 signaling may contribute to respiratory virus reinfection and evasion of vaccine-elicited immune responses. These results have important implications for the design of effective vaccines against respiratory viruses.
in vivo TIM-3 neutralization
Dietze, K. K., et al. (2013). "Combining regulatory T cell depletion and inhibitory receptor blockade improves reactivation of exhausted virus-specific CD8+ T cells and efficiently reduces chronic retroviral loads" PLoS Pathog 9(12): e1003798. PubMed
Chronic infections with human viruses, such as HIV and HCV, or mouse viruses, such as LCMV or Friend Virus (FV), result in functional exhaustion of CD8(+) T cells. Two main mechanisms have been described that mediate this exhaustion: expression of inhibitory receptors on CD8(+) T cells and expansion of regulatory T cells (Tregs) that suppress CD8(+) T cell activity. Several studies show that blockage of one of these pathways results in reactivation of CD8(+) T cells and partial reduction in chronic viral loads. Using blocking antibodies against PD-1 ligand and Tim-3 and transgenic mice in which Tregs can be selectively ablated, we compared these two treatment strategies and combined them for the first time in a model of chronic retrovirus infection. Blocking inhibitory receptors was more efficient than transient depletion of Tregs in reactivating exhausted CD8(+) T cells and reducing viral set points. However, a combination therapy was superior to any single treatment and further augmented CD8(+) T cell responses and resulted in a sustained reduction in chronic viral loads. These results demonstrate that Tregs and inhibitory receptors are non-overlapping factors in the maintenance of chronic viral infections and that immunotherapies targeting both pathways may be a promising strategy to treat chronic infectious diseases.
in vivo TIM-3 neutralization
Zelinskyy, G., et al. (2011). "Virus-specific CD8+ T cells upregulate programmed death-1 expression during acute friend retrovirus infection but are highly cytotoxic and control virus replication" J Immunol 187(7): 3730-3737. PubMed
It was recently reported that inhibitory molecules such as programmed death-1 (PD-1) were upregulated on CD8(+) T cells during acute Friend retrovirus infection and that the cells were prematurely exhausted and dysfunctional in vitro. The current study confirms that most activated CD8(+) T cells upregulated expression of PD-1 during acute infection and revealed a dichotomy of function between PD-1(hi) and PD-1(lo) subsets. More PD-1(lo) cells produced antiviral cytokines such as IFN-gamma and TNF-alpha, whereas more PD-1(hi) cells displayed characteristics of cytotoxic effectors such as production of granzymes and surface expression of CD107a. Importantly, CD8(+) T cells mediated rapid in vivo cytotoxicity and were critical for control of acute Friend virus replication. Thus, direct ex vivo analyses and in vivo experiments revealed high CD8(+) T cell functionality and indicate that PD-1 expression during acute infection is not a marker of T cell exhaustion.