InVivoMAb anti-mouse 4-1BB (CD137)
Product Details
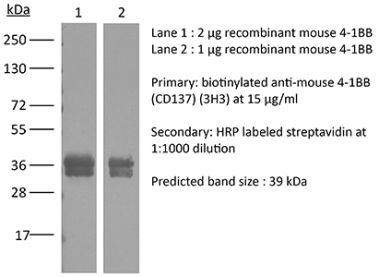
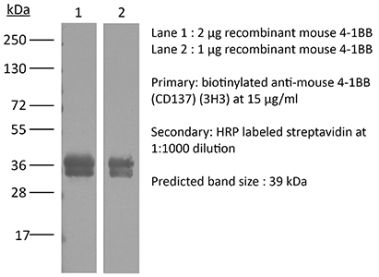
The 17B5 monoclonal antibody reacts with mouse 4-1BB, a TNF receptor superfamily member also known as CD137. 4-1BB is a 39 kDa transmembrane protein expressed by T lymphocytes, NK cells, dendritic cells, granulocytes, and mast cells. Upon binding its ligand 4-1BBL, 4-1BB provides costimulatory signals to both CD4 and CD8 T cells through the activation of NF-κB, c-Jun and p38 downstream pathways. The importance of the 4-1BB pathway has been underscored in several diseases, including cancer. The 17B5 antibody has been shown to block 4-1BB-mediated T cell proliferation in vitro.Specifications
| Isotype | Syrian hamster IgG |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb polyclonal Syrian hamster IgG |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Not available or unknown |
| Reported Applications |
in vitro 4-1BB blockade Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein A |
| RRID | AB_2687819 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vitro 4-1BB blockade
Taechangam, N., et al. (2019). "Mechanisms utilized by feline adipose-derived mesenchymal stem cells to inhibit T lymphocyte proliferation" Stem Cell Res Ther 10(1): 188. PubMed
BACKGROUND: Feline adipose-derived mesenchymal stem cells (ASCs) have been successfully used in clinical trials for the treatment of immune-mediated diseases with T cell dysregulation. However, the immunomodulatory pathways utilized by feline ASCs to suppress T cell activation have not been fully determined. We investigated the mechanisms used by feline ASCs to inhibit T cell proliferation, including the soluble factors and the cell-cell contact ligands responsible for ASC-T cell interaction. METHODS: The immunomodulatory activity of feline ASCs was evaluated via cell cycle analysis and in vitro mixed leukocyte reaction using specific immunomodulatory inhibitors. Cell-cell interactions were assessed with static adhesion assays, also with inhibitors. RESULTS: Feline ASCs decrease T cell proliferation by causing cell cycle arrest in G0-G1. Blocking prostaglandin (PGE(2)), but not IDO, partially restored lymphocyte proliferation. Although PDL-1 and CD137L are both expressed on activated feline ASCs, only the interaction of intercellular adhesion molecule 1 (ICAM-1, CD54) with its ligand, lymphocyte function-associated antigen 1 (LFA-1, CD11a/CD18), was responsible for ASC-T cell adhesion. Blocking this interaction reduced cell-cell adhesion and mediator (IFN-γ) secretion and signaling. CONCLUSIONS: Feline ASCs utilize PGE(2) and ICAM-1/LFA-1 ligand interaction to inhibit T cell proliferation with a resultant cell cycle arrest in G0-G1. These data further elucidate the mechanisms by which feline ASCs interact with T cells, help define appropriate T cell-mediated disease targets in cats that may be amenable to ASC therapy, and may also inform potential translational models for human diseases.
Flow Cytometry
Azpilikueta, A., et al. (2016). "Successful Immunotherapy against a Transplantable Mouse Squamous Lung Carcinoma with Anti-PD-1 and Anti-CD137 Monoclonal Antibodies" J Thorac Oncol 11(4): 524-536. PubMed
INTRODUCTION: Anti-programmed cell death 1 (anti-PD-1) and anti-programmed cell death ligand 1 (PD-L1) antagonist monoclonal antibodies (mAbs) against metastatic non-small cell lung cancer with special efficacy in patients with squamous cell lung cancer are being developed in the clinic. However, robust and reliable experimental models to test immunotherapeutic combinations in squamous lung tumors are still lacking. METHODS: We generated a transplantable squamous cell carcinoma cell line (UN-SCC680AJ) from a lung tumor induced by chronic N-nitroso-tris-chloroethylurea mutagenesis in A/J mice. Tumor cells expressed cytokeratins, overexpressed p40, and lacked thyroid transcription factor 1, confirming the squamous lineage reported by histological analysis. More than 200 mutations found in its exome suggested potential for antigenicity. Immunocompetent mice subcutaneously implanted with this syngeneic cell line were treated with anti-CD137 and/or anti-PD-1 mAbs and monitored for tumor growth/progression or assessed for intratumoral leukocyte infiltration using immunohistochemical analysis and flow cytometry. RESULTS: In syngeneic mice, large 12-day-established tumors derived from the transplantable cell line variant UN-SCC680AJ were amenable to curative treatment with anti-PD-1, anti-PD-L1, or anti-CD137 immunostimulatory mAbs. Single-agent therapies lost curative efficacy when treatment was started beyond day +17, whereas a combination of anti-PD-1 plus anti-CD137 achieved complete rejections. Tumor cells expressed weak baseline PD-L1 on the plasma membrane, but this could be readily induced by interferon-gamma. Combined treatment efficacy required CD8 T cells and induced a leukocyte infiltrate in which T lymphocytes co-expressing CD137 and PD-1 were prominent. CONCLUSIONS: These promising results advocate the use of combined anti-PD-1/PD-L1 plus anti-CD137 mAb immunotherapy for the treatment of squamous non-small cell lung cancer in the clinical setting.
Flow Cytometry
Eun, S. Y., et al. (2015). "4-1BB ligand signaling to T cells limits T cell activation" J Immunol 194(1): 134-141. PubMed
4-1BB ligand (4-1BBL) and its receptor, 4-1BB, are both induced on T cells after activation, but little is known about the role of 4-1BBL. In this study we show that 4-1BBL can transmit signals that limit T cell effector activity under tolerogenic conditions. Cross-linking 4-1BBL inhibited IL-2 production in vitro, primarily with suboptimal TCR stimulation. Furthermore, naive 4-1BBL-deficient OT-II transgenic T cells displayed a greater conversion to effector T cells in vivo when responding to soluble OVA peptide in wild-type hosts, whereas development of Foxp3(+) regulatory T cells was not altered. A greater number of effector T cells also differentiated from naive wild-type OT-II T cells when transferred into 4-1BB-deficient hosts, suggesting that APC-derived 4-1BB is likely to trigger 4-1BBL. Indeed, effector T cells that could not express 4-1BBL accumulated in larger numbers in vitro when stimulated with 4-1BB-expressing mesenteric lymph node dendritic cells. 4-1BBL was expressed on T cells when Ag presentation was limiting, and 4-1BBL was aberrantly expressed at very high levels on T cells that could not express 4-1BB. Trans-ligation, Ab capture, and endocytosis experiments additionally showed that T cell-intrinsic 4-1BB regulated internalization of membrane 4-1BBL, implying that the strong induction of 4-1BB on T cells may counteract the suppressive function of 4-1BBL by limiting its availability. These data suggest that 4-1BBL expressed on T cells can restrain effector T cell development, creating a more favorable regulatory T cell to effector cell balance under tolerogenic conditions, and this may be particularly active in mucosal barrier tissues where 4-1BB-expressing regulatory dendritic cells present Ag.
Flow Cytometry
Fuse, S., et al. (2007). "CD8+ T cell dysfunction and increase in murine gammaherpesvirus latent viral burden in the absence of 4-1BB ligand" J Immunol 178(8): 5227-5236. PubMed
Studies of costimulatory receptors belonging to the TNFR family have revealed their diverse roles in affecting different stages of the T cell response. The 4-1BB ligand (4-1BBL)/4-1BB pathway has emerged as a receptor-ligand pair that impacts not the initial priming, but later phases of the T cell response, such as sustaining clonal expansion and survival, maintaining memory CD8(+) T cells, and supporting secondary expansion upon Ag challenge. Although the role of this costimulatory pathway in CD8(+) T cell responses to acute viral infections has been well-studied, its role in controlling chronic viral infections in vivo is not known to date. Using the murine gammaherpesvirus-68 (MHV-68) model, we show that 4-1BBL-deficient mice lack control of MHV-68 during latency and show significantly increased latent viral loads. In contrast to acute influenza infection, the numbers of MHV-68-specific memory CD8(+) T cells were maintained during latency. However, the virus-specific CD8(+) T cells showed defects in function, including decreased cytolytic function and impaired secondary expansion. Thus, 4-1BBL deficiency significantly affects the function, but not the number, of virus-specific CD8(+) T cells during gammaherpesvirus latency, and its absence results in an increased viral burden. Our study suggests that the 4-1BB costimulatory pathway plays an important role in controlling chronic viral infections.
Flow Cytometry
Nishimoto, H., et al. (2005). "Costimulation of mast cells by 4-1BB, a member of the tumor necrosis factor receptor superfamily, with the high-affinity IgE receptor" Blood 106(13): 4241-4248. PubMed
Mast cells are the major effector-cell type for immediate hypersensitivity and other forms of allergic reactions. Expression of 4-1BB, a member of the tumor necrosis factor receptor superfamily, is induced at mRNA and protein levels on stimulation through the high-affinity receptor for immunoglobulin E (IgE; FcepsilonRI). In this study, we present evidence that agonistic anti-4-1BB antibodies can enhance FcepsilonRI-induced cytokine production and secretion. Consistent with this, 4-1BB-deficient mast cells exhibit reduced degranulation and cytokine production on FcepsilonRI stimulation. Analysis of 4-1BB ligand (4-1BBL)-deficient cells supported this notion. As a potential mechanism for these defects, we identified a defect in Ca2+ flux induced by FcepsilonRI stimulation. The defective Ca2+ flux could be accounted for by the reduced activity of Lyn/Btk/phospholipase C-gamma2 pathway and constitutive interactions between 4-1BB and Lyn. Therefore, FcepsilonRI-inducible 4-1BB plays a costimulatory function together with FcepsilonRI stimulation.
in vitro 4-1BB blockade, Flow Cytometry
Zheng, G., et al. (2004). "The 4-1BB costimulation augments the proliferation of CD4+CD25+ regulatory T cells" J Immunol 173(4): 2428-2434. PubMed
The thymus-derived CD4(+)CD25(+) T cells belong to a subset of regulatory T cells potentially capable of suppressing the proliferation of pathogenic effector T cells. Intriguingly, these suppressor cells are themselves anergic, proliferating poorly to mitogenic stimulation in culture. In this study, we find that the 4-1BB costimulator receptor, best known for promoting the proliferation and survival of CD8(+) T cells, also induces the proliferation of the CD4(+)CD25(+) regulatory T cells both in culture and in vivo. The proliferating CD4(+)CD25(+) T cells produce no detectable IL-2, suggesting that 4-1BB costimulation of these cells does not involve IL-2 production. The 4-1BB-expanded CD4(+)CD25(+) T cells are functional, as they remain suppressive to other T cells in coculture. These results support the notion that the peripheral expansion of the CD4(+)CD25(+) T cells is controlled in part by costimulation.