InVivoMAb anti-human/rat HER2 (neu)
Product Details
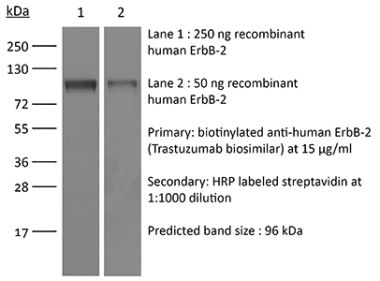
The 7.16.4 monoclonal antibody reacts with human and rat HER2 (human epidermal growth factor receptor 2) also known as neu peptide, CD340, ErbB-2, and p185. HER2 is a 185 kDa transmembrane, receptor-like glycoprotein with intrinsic tyrosine kinase activity that is part of several cell surface receptor complexes. HER2 lacks an identified ligand however, the kinase can be activated in the absence of a ligand when overexpressed. HER2 is a proto-oncoprotein that is commonly overexpressed on a variety of different tumors. Approximately 40% of human breast cancers overexpress HER2. HER2 overexpression is associated with poorer overall survival rates, shorter times to disease progression, and increased resistance to chemotherapy. Because of these clinical characteristics anti-HER2 monoclonal antibody therapy is now a standard for the treatment of advanced breast cancers that overexpress HER2. The 7.16.4 antibody has been shown to inhibit the growth of HER2-overexpressing tumors both in vitro and in vivo.Specifications
| Isotype | Mouse IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG2a isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | neu-transfected NIH 3T3 cells |
| Reported Applications |
in vivo HER2/neu inhibition in vitro HER2/neu inhibition Immunoprecipitation Immunofluorescence Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 μM filtered |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_2687800 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vivo HER2/neu inhibition
Kodumudi, K. N., et al. (2019). "Sequential Anti-PD1 Therapy Following Dendritic Cell Vaccination Improves Survival in a HER2 Mammary Carcinoma Model and Identifies a Critical Role for CD4 T Cells in Mediating the Response" Front Immunol 10: 1939. PubMed
Patients with metastatic HER2 breast cancer (MBC) often become resistant to HER 2 targeted therapy and have recurrence of disease. The Panacea trial suggested that HER2 MBC patients were more likely to respond to checkpoint therapy if TIL were present or if tumor expressed PD-L1. We assessed whether type I polarized dendritic cells (DC1) could improve checkpoint therapy in a preclinical model of HER2(+) breast cancer. TUBO bearing mice were vaccinated with either MHC class I or class II HER2 peptide pulsed DC1 (class I or class II HER2-DC1) concurrently or sequentially with administration of anti-PD-1 or anti-PDL1. Infiltration of tumors by immune cells, induction of anti-HER2 immunity and response to therapy was evaluated. Class I or class II HER2-DC1 vaccinated mice generated anti-HER2 CD8 or CD4+ T cell immune responses and demonstrated delayed tumor growth. Combining both MHC class I and II HER2-pulsed DC1 did not further result in inhibition of tumor growth or enhanced survival compared to individual administration. Interestingly class II HER2-DC1 led to both increased CD4 and CD8 T cells in the tumor microenvironment while class I peptides typically resulted in only increased CD8 T cells. Anti-PD-1 but not anti-PD-L1 administered sequentially with class I or class II HER2-DC1 vaccine could improve the efficacy of HER2-DC1 vaccine as measured by tumor growth, survival, infiltration of tumors by T cells and increase in systemic anti-HER2 immune responses. Depletion of CD4+ T cells abrogated the anti-tumor efficacy of combination therapy with class II HER2-DC1 and anti-PD-1, suggesting that tumor regression was CD4 dependent. Since class II HER2-DC1 was as effective as class I, we combined class II HER2-DC1 vaccine with anti-rat neu antibodies and anti-PD-1 therapy. Combination therapy demonstrated further delay in tumor growth, and enhanced survival compared to control mice. In summary, Class II HER2-DC1 drives both a CD4 and CD8 T cell tumor infiltration that leads to increased survival, and in combination with anti-HER2 therapy and checkpoint blockade can improve survival in preclinical models of HER2 positive breast cancer and warrants exploration in patients with HER2 MBC.
in vivo HER2/neu inhibition
Wang, Q., et al. (2019). "Single-cell profiling guided combinatorial immunotherapy for fast-evolving CDK4/6 inhibitor-resistant HER2-positive breast cancer" Nat Commun 10(1): 3817. PubMed
Acquired resistance to targeted cancer therapy is a significant clinical challenge. In parallel with clinical trials combining CDK4/6 inhibitors to treat HER2+ breast cancer, we sought to prospectively model tumor evolution in response to this regimen in vivo and identify a clinically actionable strategy to combat drug resistance. Despite a promising initial response, acquired resistance emerges rapidly to the combination of anti-HER2/neu antibody and CDK4/6 inhibitor Palbociclib. Using high-throughput single-cell profiling over the course of treatments, we reveal a distinct immunosuppressive immature myeloid cell (IMC) population to infiltrate the resistant tumors. Guided by single-cell transcriptome analysis, we demonstrate that combination of IMC-targeting tyrosine kinase inhibitor cabozantinib and immune checkpoint blockade enhances anti-tumor immunity, and overcomes the resistance. Furthermore, sequential combinatorial immunotherapy enables a sustained control of the fast-evolving CDK4/6 inhibitor-resistant tumors. Our study demonstrates a translational framework for treating rapidly evolving tumors through preclinical modeling and single-cell analyses.
in vivo HER2/neu inhibition
Park, S., et al. (2010). "The therapeutic effect of anti-HER2/neu antibody depends on both innate and adaptive immunity" Cancer Cell 18(2): 160-170. PubMed
Anti-HER2/neu antibody therapy is reported to mediate tumor regression by interrupting oncogenic signals and/or inducing FcR-mediated cytotoxicity. Here, we demonstrate that the mechanisms of tumor regression by this therapy also require the adaptive immune response. Activation of innate immunity and T cells, initiated by antibody treatment, was necessary. Intriguingly, the addition of chemotherapeutic drugs, although capable of enhancing the reduction of tumor burden, could abrogate antibody-initiated immunity leading to decreased resistance to rechallenge or earlier relapse. Increased influx of both innate and adaptive immune cells into the tumor microenvironment by a selected immunotherapy further enhanced subsequent antibody-induced immunity, leading to increased tumor eradication and resistance to rechallenge. This study proposes a model and strategy for anti-HER2/neu antibody-mediated tumor clearance.
in vivo HER2/neu inhibition, in vitro HER2/neu inhibition
Knutson, K. L., et al. (2004). "Neu antigen-negative variants can be generated after neu-specific antibody therapy in neu transgenic mice" Cancer Res 64(3): 1146-1151. PubMed
Prolonged administration of HER-2/neu-specific monoclonal antibody therapy is now widely used for the treatment of HER-2/neu-overexpressing tumors in advanced-stage breast cancer patients. Monoclonal antibody therapy has the potential to promote reduced tumor expression of HER-2/neu by receptor down-modulation and/or the generation of antigen-negative variants. Loss of antigen by either mechanism could potentially impact subsequent therapeutic strategies targeting HER-2/neu. In this study, the effects of chronic neu-specific monoclonal antibody therapy on tumor growth and neu protein expression were examined in a murine model of neu-overexpressing breast cancer. Treatment of neu-overexpressing tumors with neu-specific antibody, in vitro or in vivo, resulted in significant tumor growth inhibition. When neu antibody was used to treat neu-overexpressing tumor cells both in vitro and in vivo in tumor-bearing mice, neu receptor expression was not diminished after cessation of therapy. However, in the setting of clinically undetectable disease in a fraction of animals, antigen-negative variants were generated. An understanding of the effects of monoclonal antibodies on target antigen expression is critical for the future design and testing of novel HER-2/neu-targeted therapies administered in combination with or after HER-2/neu-specific monoclonal antibody therapy.
in vitro HER2/neu inhibition, Flow Cytometry, Immunoprecipitation
Zhang, H., et al. (1999). "Shared antigenic epitopes and pathobiological functions of anti-p185(her2/neu) monoclonal antibodies" Exp Mol Pathol 67(1): 15-25. PubMed
We have studied two anti-p185 antibodies: the monoclonal antibody 7. 16.4 and rhuMAb 4D5, which were raised against the the ectodomain of rat (p185(neu)), and the human (p185(her2/neu)) homolog, respectively. Studies on the structure of these two antibodies indicate that they share structural similarity in the variable region, especially the CDR3 region, which determines the antibody-antigen interaction. Further studies by flow cytometry revealed that 7.16.4 can compete with rhuMAb4D5 for binding to the cell surface p185(her2/neu), suggesting that these two antibodies share an epitope on the p185 receptor. Furthermore, 7.16.4 can also inhibit proliferation and transformation caused by p185(her2/neu). Moreover the rhuMAb 4D5 binds to the rat p185(neu). With the observation that 7.16.4 positively stains human breast cancer tissues that overexpress p185(her2/neu), 7.16.4 may be useful for the pathological diagnosis and therapy of human tumors.
in vivo HER2/neu inhibition
Drebin, J. A., et al. (1986). "Inhibition of tumor growth by a monoclonal antibody reactive with an oncogene-encoded tumor antigen" Proc Natl Acad Sci U S A 83(23): 9129-9133. PubMed
The neu oncogene encodes a 185-kDa transmembrane glycoprotein tumor antigen, termed p185. We have recently described a monoclonal antibody reactive with a cell surface domain of the p185 molecule. In vivo treatment with this anti-p185 monoclonal antibody was able to significantly inhibit the tumorigenic growth of neu-transformed NIH 3T3 cells implanted into nude mice. Such treatment had no effect on the tumorigenic growth of Ha-ras-transformed NIH 3T3 cells. Furthermore, anti-p185 antibody treatment was able to inhibit the growth of the rat neuroblastoma cells from which the neu oncogene was initially isolated. These results demonstrate that a monoclonal antibody reactive with the extracellular domain of an oncogene-encoded protein can exert a significant antitumor effect; such antibodies may prove useful in the therapy of certain malignancies.
Flow Cytometry, Immunoprecipitation, Immunofluorescence
Drebin, J. A., et al. (1984). "Monoclonal antibodies identify a cell-surface antigen associated with an activated cellular oncogene" Nature 312(5994): 545-548. PubMed
A variety of antigens have been identified on the surface of the malignant cell. However, identical antigens are often found on non-malignant cells of the same or different histological origin, or of a different stage of embryonic development. Many of these tumour-associated antigens appear to be only incidentally expressed on neoplastic cells. Clearly, it would be of great interest to identify cell-surface antigens whose expression is associated specifically with the transformed state and linked directly with the mechanisms responsible for transformation. The detection of activated cellular oncogenes in human and animal cancer cells by the technique of DNA transfection has allowed the isolation of genetic elements which are thought to have a critical role in malignancy. Here, in an effort to identify cell-surface antigens associated with the neoplastic process, we have generated hybridomas which secrete monoclonal antibodies that react specifically with cell-surface determinants found on NIH 3T3 cells transformed by transfection with a group of rat neuroblastoma oncogenes. These antibodies bind to and immunoprecipitate a phosphoprotein of relative molecular mass 185,000 (185 K) from a DNA donor rat neuroblastoma and 13 independent rat neuroblastoma DNA transfectants. There was no antibody reactivity with normal NIH 3T3 cells or with NIH 3T3 cells transformed by various other agents.