InVivoMAb anti-human CD71 (TfR)
Product Details
The 5E9C11 monoclonal antibody reacts with human CD71 also known as the transferrin receptor. CD71 is a type II homodimeric transmembrane glycoprotein which is expressed on the surface of proliferating cells, reticulocytes, and erythroid precursors. CD71 plays a role in the control of cellular proliferation and is required for iron import from transferrin into cells by endocytosis. CD71 is expressed on malignant cells at high levels and its expression correlates with cancer progression. This high expression on malignant cells along with CD71’s ability to internalize and the necessity of iron for cancer cell proliferation makes the transferrin receptor an attractive target to exploit for the delivery of drugs into malignant cells. The 5E9C11 antibody does not inhibit the binding of transferrin. This antibody has been previously used to deliver immunotoxins to human tumor xenografts in vivo in mice.Specifications
| Isotype | Mouse IgG1, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG1 isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Human acute lymphocytic leukemia T cell line HSB-2 |
| Reported Applications |
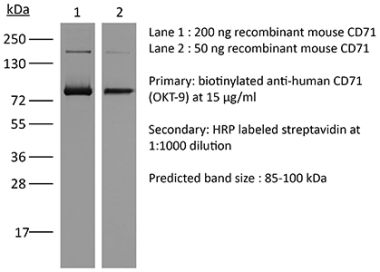
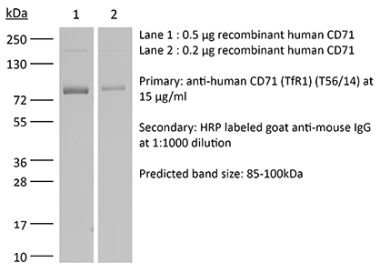
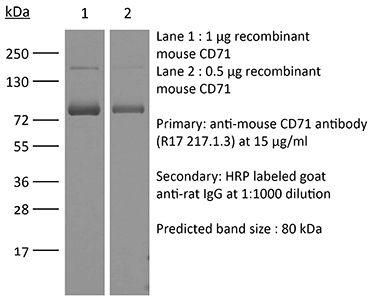
Western blot Immunoprecipitation Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 μM filtered |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_2894762 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
Immunoprecipitation, Western Blot
Denti, S., et al. (2004). "RanBPM is a phosphoprotein that associates with the plasma membrane and interacts with the integrin LFA-1" J Biol Chem 279(13): 13027-13034. PubMed
Integrin adhesion receptors can act as signaling receptors that transmit information from the extracellular environment to the interior of the cell, affecting many fundamental cellular processes, such as cell motility, proliferation, differentiation, and survival. Integrin signaling depends on the formation of organized sub-membrane complexes that comprise cytoskeletal, adapter, and signaling molecules. The identification of molecules that interact with the cytoplasmic domain of integrins has been the focus of research aimed to elucidating the mechanistic basis of integrin signal transduction. We have identified RanBPM as a novel interactor of the beta(2) integrin LFA-1 in a yeast-two-hybrid screen. In the same assay, RanBPM also interacted with the beta(1) integrin cytoplasmic domain. We demonstrate that RanBPM is a peripheral membrane protein and that integrins and RanBPM interact in vitro and in vivo and co-localize at the cell membrane. We find that RanBPM is phosphorylated on serine residues; phosphorylation of RanBPM is increased by stress stimuli and decreased by treatment with the p38 kinase inhibitor SB203580. Transfection of RanBPM synergizes with LFA-1-mediated adhesion in the transcriptional activation of an AP-1-dependent promoter, indicating that the two proteins interact functionally as well. We suggest that RanBPM may constitute a molecular scaffold that contributes to coupling LFA-1 and other integrins with intracellular signaling pathways.
Flow Cytometry
Hmama, Z., et al. (1998). "Attenuation of HLA-DR expression by mononuclear phagocytes infected with Mycobacterium tuberculosis is related to intracellular sequestration of immature class II heterodimers" J Immunol 161(9): 4882-4893. PubMed
MHC class II expression was examined in macrophages infected with Mycobacterium tuberculosis. IFN-gamma increased the surface expression of class II molecules in THP-1 cells and this was markedly reduced in cells infected with M. tuberculosis. Despite this effect, steady state levels of HLA-DRalpha, HLA-DRbeta, and invariant (Ii) chains were equivalent in control and infected cells. Metabolic labeling combined with pulse-chase experiments and biochemical analysis showed that the majority of class II molecules in infected cells became resistant to endoglycosidase H, consistent with normal Golgi processing. However, results of intracellular staining and dual color confocal microscopy revealed a significant defect in transport of newly synthesized class II molecules through the endocytic compartment. Thus, compared with findings in control cells, class II molecules in infected cells colocalized to a minimal extent with a lysosomal-associated membrane protein-1+ endosomal compartment. In addition, in contrast to control cells, class II molecules in infected cells failed to colocalize with endocytosed BSA under conditions where this marker is known to label late endosomes, lysosomes, and the MHC class II compartment. Consistent with defective transport along the endocytic pathway, the maturation of SDS-stable class II alphabeta dimers–dependent upon removal of Ii chain and peptide loading of class II dimers in the MHC class II compartment–was markedly impaired in M. tuberculosis-infected cells. These findings indicate that defective transport and processing of class II molecules through the endosomal/lysosomal system is responsible for diminished cell surface expression of MHC class II molecules in cells infected with M. tuberculosis.
Scott, C. F., Jr., et al. (1987). "An immunotoxin composed of a monoclonal antitransferrin receptor antibody linked by a disulfide bond to the ribosome-inactivating protein gelonin: potent in vitro and in vivo effects against human tumors" J Natl Cancer Inst 79(5): 1163-1172. PubMed
An immunoconjugate was prepared containing a disulfide linker between a murine monoclonal antibody (5E9), which recognized the human transferrin receptor, and the ribosome-inactivating protein gelonin. This immunoconjugate was found to consist of two major species, 5E9-gelonin2 and 5E9-gelonin1, and a minor species of 5E9-gelonin3 and less than 10% of either free antibody or gelonin. 5E9-gelonin was extremely toxic in vitro to human tumor cell lines expressing the 5E9 antigen, including a Burkitt’s lymphoma, an adult T-cell acute lymphocytic leukemia, an acute myelogenous leukemia, a promyelocytic leukemia, and a cervical carcinoma line. A 24-hour exposure to 10(-9) M immunoconjugate killed 90-99.9% of tumor cells, depending on the cell line. A 5E9-negative murine leukemia was not sensitive to this conjugate. Pharmacokinetic analysis of the disappearance of this immunoconjugate from the murine circulation revealed that it had a biphasic clearance, with an initial rapid phase with a half-life (t1/2) of 3 hours and a later, slower phase with a t1/2 of about 1 day. Analysis of blood samples by sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis revealed that a substantial degree of disulfide-linker breakdown occurred in vivo and that the 5E9-gelonin2 species was cleared more rapidly than the 5E9-gelonin1. With use of the same clonogenic assays used to measure in vitro toxicity, biologically active immunoconjugate could be detected in murine plasma for up to 24 hours after iv administration, but the concentration of immunoconjugate by this measure was considerably less than that predicted by SDS-gel electrophoresis. The ability to deliver immunoconjugate to tumor cells in vivo was studied with use of the Burkitt’s lymphoma Namalwa as a xenograft in nude mice. It was possible to deliver substantial amounts of immunoconjugate to Namalwa cells in xenografted ascites with direct ip inoculation; lower but significant amounts of immunoconjugate could be delivered to this xenograft after systemic iv administration, provided the tumor burden was low. The 5E9-gelonin conjugate, when administered iv at the time of ip tumor inoculation, prolonged survival of nude mice bearing Namalwa or other human tumors as ascites xenografts and delayed or prevented the growth of subcutaneous nodules of Namalwa in an antigen-specific fashion after a single iv injection. Direct intratumoral administration also inhibited the growth of visible subcutaneous nodules of Namalwa. This immunoconjugate may be useful in the treatment of human cancer.