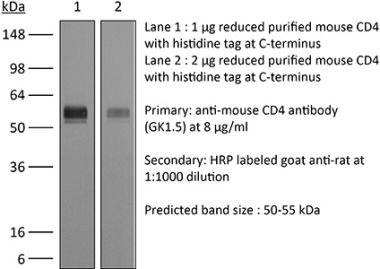
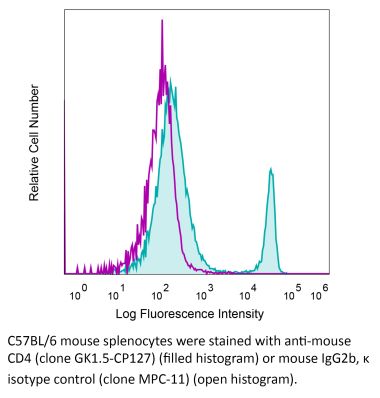
InVivoMAb anti-human CD4
Product Details
The OKT-4 monoclonal antibody reacts with the human CD4. The CD4 antigen is a 55 kDa cell surface type I membrane glycoprotein belonging to the immunoglobulin superfamily. CD4 acts as a co-receptor which in cooperation with the T cell receptor (TCR) interacts with class II MHC molecules displayed by antigen presenting cells (APC). CD4 is expressed by the majority of thymocytes, most helper T cells, a subset of NK-T cells and weakly by dendritic cells and macrophages. CD4 plays an important role in the development of T cells and is required for mature T cells to function optimally.Specifications
| Isotype | Mouse IgG2b, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG2b isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Human peripheral T cells |
| Reported Applications |
in vitro T cell stimulation/activation in vivo CD4+ T cell depletion in humanized mice Flow cytometry Immunoprecipitation |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
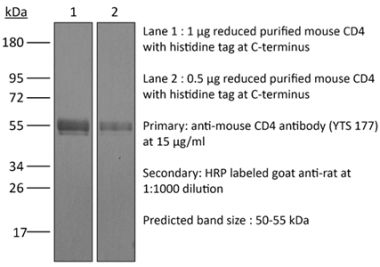
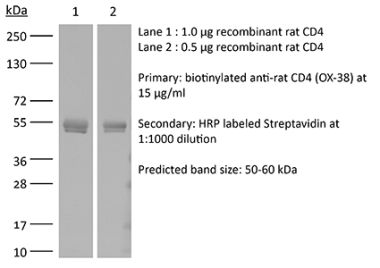
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107638 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vivo CD4+ T cell depletion in humanized mice
Danzer, H., et al. (2020). "Human Fcγ-receptor IIb modulates pathogen-specific versus self-reactive antibody responses in lyme arthritis" Elife 9 . PubMed
Pathogen-specific antibody responses need to be tightly regulated to generate protective but limit self-reactive immune responses. While loss of humoral tolerance has been associated with microbial infections, the pathways involved in balancing protective versus autoreactive antibody responses in humans are incompletely understood. Studies in classical mouse model systems have provided evidence that balancing of immune responses through inhibitory receptors is an important quality control checkpoint. Genetic differences between inbred mouse models and the outbred human population and allelic receptor variants not present in mice; however, argue for caution when directly translating these findings to the human system. By studying Borrelia burgdorferi infection in humanized mice reconstituted with human hematopoietic stem cells from donors homozygous for a functional or a non-functional FcγRIIb allele, we show that the human inhibitory FcγRIIb is a critical checkpoint balancing protective and autoreactive immune responses, linking infection with induction of autoimmunity in the human immune system.
in vivo CD4+ T cell depletion in humanized mice
Homet Moreno, B., et al. (2016). "Response to Programmed Cell Death-1 Blockade in a Murine Melanoma Syngeneic Model Requires Costimulation, CD4, and CD8 T Cells" Cancer Immunol Res 4(10): 845-857. PubMed
The programmed cell death protein 1 (PD-1) limits effector T-cell functions in peripheral tissues, and its inhibition leads to clinical benefit in different cancers. To better understand how PD-1 blockade therapy modulates the tumor-host interactions, we evaluated three syngeneic murine tumor models, the BRAF(V600E)-driven YUMM1.1 and YUMM2.1 melanomas, and the carcinogen-induced murine colon adenocarcinoma MC38. The YUMM cell lines were established from mice with melanocyte-specific BRAF(V600E) mutation and PTEN loss (BRAF(V600E)/PTEN(-/-)). Anti-PD-1 or anti-PD-L1 therapy engendered strong antitumor activity against MC38 and YUMM2.1, but not YUMM1.1. PD-L1 expression did not differ between the three models at baseline or upon interferon stimulation. Whereas mutational load was high in MC38, it was lower in both YUMM models. In YUMM2.1, the antitumor activity of PD-1 blockade had a critical requirement for both CD4 and CD8 T cells, as well as CD28 and CD80/86 costimulation, with an increase in CD11c(+)CD11b(+)MHC-II(high) dendritic cells and tumor-associated macrophages in the tumors after PD-1 blockade. Compared with YUMM1.1, YUMM2.1 exhibited a more inflammatory profile by RNA sequencing analysis, with an increase in expression of chemokine-trafficking genes that are related to immune cell recruitment and T-cell priming. In conclusion, response to PD-1 blockade therapy in tumor models requires CD4 and CD8 T cells and costimulation that is mediated by dendritic cells and macrophages.
in vitro T cell stimulation/activation
Klammt, C., et al. (2015). "T cell receptor dwell times control the kinase activity of Zap70" Nat Immunol 16(9): 961-969. PubMed
Kinase recruitment to membrane receptors is essential for signal transduction. However, the underlying regulatory mechanisms are poorly understood. We investigated how conformational changes control T cell receptor (TCR) association and activity of the kinase Zap70. Structural analysis showed that TCR binding or phosphorylation of Zap70 triggers a transition from a closed, autoinhibited conformation to an open conformation. Using Zap70 mutants with defined conformations, we found that TCR dwell times controlled Zap70 activity. The closed conformation minimized TCR dwell times and thereby prevented activation by membrane-associated kinases. Parallel recruitment of coreceptor-associated Lck kinase to the TCR ensured Zap70 phosphorylation and stabilized Zap70 TCR binding. Our study suggests that the dynamics of cytosolic enzyme recruitment to the plasma membrane regulate the activity and function of receptors lacking intrinsic catalytic activity.
in vivo CD4+ T cell depletion in humanized mice
Billerbeck, E., et al. (2013). "Characterization of human antiviral adaptive immune responses during hepatotropic virus infection in HLA-transgenic human immune system mice" J Immunol 191(4): 1753-1764. PubMed
Humanized mice have emerged as a promising model to study human immunity in vivo. Although they are susceptible to many pathogens exhibiting an almost exclusive human tropism, human immune responses to infection remain functionally impaired. It has recently been demonstrated that the expression of HLA molecules improves human immunity to lymphotropic virus infections in humanized mice. However, little is known about the extent of functional human immune responses in nonlymphoid tissues, such as in the liver, and the role of HLA expression in this context. Therefore, we analyzed human antiviral immunity in humanized mice during a hepatotropic adenovirus infection. We compared immune responses of conventional humanized NOD SCID IL-2Rgamma-deficient (NSG) mice to those of a novel NOD SCID IL-2Rgamma-deficient strain transgenic for both HLA-A*0201 and a chimeric HLA-DR*0101 molecule. Using a firefly luciferase-expressing adenovirus and in vivo bioluminescence imaging, we demonstrate a human T cell-dependent partial clearance of adenovirus-infected cells from the liver of HLA-transgenic humanized mice. This correlated with liver infiltration and activation of T cells, as well as the detection of Ag-specific humoral and cellular immune responses. When infected with a hepatitis C virus NS3-expressing adenovirus, HLA-transgenic humanized mice mounted an HLA-A*0201-restricted hepatitis C virus NS3-specific CD8(+) T cell response. In conclusion, our study provides evidence for the generation of partial functional antiviral immune responses against a hepatotropic pathogen in humanized HLA-transgenic mice. The adenovirus reporter system used in our study may serve as simple in vivo method to evaluate future strategies for improving human intrahepatic immune responses in humanized mice.
Flow Cytometry, Immunoprecipitation
Hepburn, T. W., et al. (2003). "Antibody-mediated stripping of CD4 from lymphocyte cell surface in patients with rheumatoid arthritis" Rheumatology (Oxford) 42(1): 54-61. PubMed
Kinase recruitment to membrane receptors is essential for signal transduction. However, the underlying regulatory mechanisms are poorly understood. We investigated how conformational changes control T cell receptor (TCR) association and activity of the kinase Zap70. Structural analysis showed that TCR binding or phosphorylation of Zap70 triggers a transition from a closed, autoinhibited conformation to an open conformation. Using Zap70 mutants with defined conformations, we found that TCR dwell times controlled Zap70 activity. The closed conformation minimized TCR dwell times and thereby prevented activation by membrane-associated kinases. Parallel recruitment of coreceptor-associated Lck kinase to the TCR ensured Zap70 phosphorylation and stabilized Zap70 TCR binding. Our study suggests that the dynamics of cytosolic enzyme recruitment to the plasma membrane regulate the activity and function of receptors lacking intrinsic catalytic activity.