InVivoMAb anti-human CD25 (IL-2Rα)
Product Details
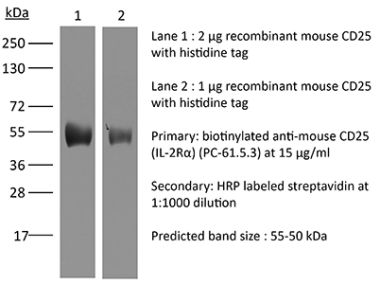
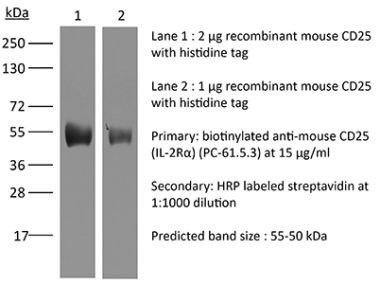
The 7G7B6 monoclonal antibody reacts with human IL-2Rα also known as CD25, Ly-43, p55, or Tac. IL-2Rα is the 55 kDa ligand-binding subunit of the interleukin 2 receptor alpha chain. IL-2Rα is expressed on activated mature T and B lymphocytes, thymocyte subsets, pre-B cells, and T regulatory cells. IL-2Rα has been shown to play roles in lymphocyte differentiation, activation, and proliferation. Alone, the IL-2Rα binds IL-2 with relatively low affinity however, when IL-2Rα associates with IL-2Rβ (CD122) and the common gamma chain (CD132) the complex binds IL-2 with high affinity.Specifications
| Isotype | Mouse IgG2a |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG2a isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Human PBMCs |
| Reported Applications |
in vivo regulatory T cell depletion in humanized mice Immunoprecipitation Immunofluorescence |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107617 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vivo regulatory T cell depletion in humanized mice
Li, Y., et al. (2017). "Regulatory T cells control toxicity in a humanized model of IL-2 therapy" Nat Commun 8(1): 1762. PubMed
While patient selection and clinical management have reduced high-dose IL-2 (HDIL2) immunotherapy toxicities, the immune mechanisms that underlie HDIL2-induced morbidity remain unclear. Here we show that dose-dependent morbidity and mortality of IL-2 immunotherapy can be modeled in human immune system (HIS) mice. Depletion of human T cell subsets during the HDIL2 treatment reduces toxicity, pointing to the central function of T cells. Preferential expansion of effector T cells secondary to defective suppressive capacity of regulatory T (T(reg)) cells after HDIL2 therapy further underscores the importance of T(reg) in the maintenance of immune tolerance. IL-2 toxicity is induced by selective depletion or inhibition of T(reg) after LDIL2 therapy, and is ameliorated in HDIL2-treated HIS mice receiving the PIM-1 kinase inhibitor, Kaempferol. Modeling IL-2 pathophysiology in HIS mice offers a means to understand the functions of effector and regulatory T cells in immune-mediated toxicities associated with cancer immunotherapy.
Immunoprecipitation
Hong, X., et al. (2015). "SAP97 blocks the RXR ER retention signal of NMDA receptor subunit GluN1-3 through its SH3 domain" Biochim Biophys Acta 1853(2): 489-499. PubMed
Plakophilin-1 (PKP-1) is an armadillo family protein critical for desmosomal adhesion and epidermal integrity. In the autoimmune skin-blistering disease pemphigus vulgaris (PV), autoantibodies (IgG) target the desmosomal cadherin desmoglein 3 (Dsg3) and compromise keratinocyte cell-cell adhesion. Here, we report that enhanced expression of PKP-1 protects keratinocytes from PV IgG-induced loss of cell-cell adhesion. PKP-1 prevents loss of Dsg3 and other desmosomal proteins from cell-cell borders and prevents alterations in desmosome ultrastructure in keratinocytes treated with PV IgG. Using a series of Dsg3 chimeras and deletion constructs, we find that PKP-1 clusters Dsg3 with the desmosomal plaque protein desmoplakin in a manner dependent on the plakoglobin-binding domain of the Dsg3 tail. Furthermore, PKP-1 expression transforms desmosome adhesion from a calcium-dependent to a calcium-independent and hyperadhesive state. These results demonstrate that manipulating the expression of a single desmosomal plaque protein can block the pathogenic effects of PV IgG on keratinocyte adhesion.
Immunofluorescence
Tucker, D. K., et al. (2014). "Plakophilin-1 protects keratinocytes from pemphigus vulgaris IgG by forming calcium-independent desmosomes" J Invest Dermatol 134(4): 1033-1043. PubMed
SAP97 is directly involved in exporting NMDA receptors with a specific subunit composition from the endoplasmic reticulum (ER). Characterization of the interactions between SAP97 and an NMDA receptor splice variant, GluN1-3, and of the effects on forward trafficking revealed that an ER-level interaction blocked the RXR ER-retention motif in the GluN1-3 cytoplasmic C-terminus in the context of both reporter molecules and full-length receptors. Binding of SAP97 to the PDZ-binding domain of GluN1-3 was required, but the blockade of ER-retention was mediated by the SH3-GuK domains coupled with the action of the N-terminus of SAP97. While other domains of SAP97 were involved in forward trafficking of GluN1-3 out of the ER, the SH3 domain was necessary and sufficient to block the ER retention. This is the first direct evidence for the masking of ER-retention signals by PDZ domain-containing proteins, and provides detailed underlying mechanistic requirements. Such a mechanism could be central to modulating the ER exit of receptors into local, non-conventional or conventional, secretory pathways in neurons.