InVivoMAb anti-human IFNγ
Product Details
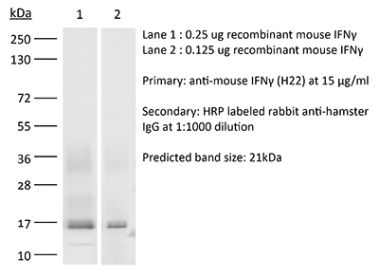
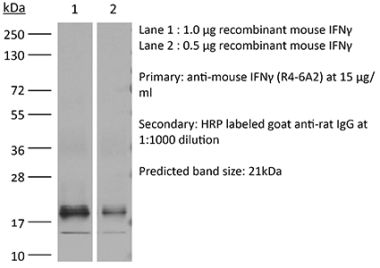
The B133.5 monoclonal antibody reacts with human IFNγ (interferon gamma) a 20 kDa soluble pleiotropic cytokine and the sole member of the type II class of interferons. IFNγ is primarily produced by activated lymphocytes including T, B, NK cells, and ILCs. IFNγ exerts immunoregulatory, anti-proliferative, anti-viral, and proinflammatory activities and plays an important role in activation, growth, and differentiation of T and B lymphocytes, macrophages, NK cells and other non-hematopoietic cell types. Additionally, IFNγ induces the production of cytokines, Fc receptor, and adhesion molecules and up-regulates MHC class I and II antigen expression by antigen presenting cells during an immune response. IFNγ has also been shown to modulate macrophage effector functions, influence isotype switching and induce the secretion of immunoglobulins by B cells. IFNγ signals through the IFN gamma receptor which exists as a heterodimer composed of CD119 (IFN gamma receptor 1) and AF-1 (IFN gamma receptor 2). The IFNγ receptor is expressed ubiquitously on almost all cell types with the exception of mature erythrocytes. The B133.5 antibody is a neutralizing antibody.Specifications
| Isotype | Mouse IgG1 |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG1 isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Recombinant human IFNγ |
| Reported Applications |
in vivo IFNγ neutralization in humanized mice in vitro IFNγ neutralization |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein A |
| RRID | AB_2687717 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Recommended Products
in vivo IFNγ neutralization in humanized mice
Cremasco, F., et al. (2021). "Cross-linking of T cell to B cell lymphoma by the T cell bispecific antibody CD20-TCB induces IFNγ/CXCL10-dependent peripheral T cell recruitment in humanized murine model" PLoS One 16(1): e0241091. PubMed
Diffuse large B cell lymphomas (DLBCL) are a highly heterogeneous subtype of Non Hodgkin Lymphoma (NHL), accounting for about 25% of NHL. Despite an increased progression-free survival upon therapy, 40-50% of patients develop relapse/refractory disease, therefore there remains an important medical need. T cell recruiting therapies, such as the CD20xCD3 T cell bi-specific antibody CD20-TCB (RG6026 or glofitamab), represent a novel approach to target all stages of DLBCL, especially those that fail to respond to multiple lines of treatment. We aimed for a better understanding of the molecular features related to the mode of action (MoA) of CD20-TCB in inducing Target/T cell synapse formation and human T cell recruitment to the tumor. To directly evaluate the correlation between synapse, cytokine production and anti-tumor efficacy using CD20-TCB, we developed an innovative preclinical human DLBCL in vivo model that allowed tracking in vivo human T cell dynamics by multiphoton intravital microscopy (MP-IVM). By ex vivo and in vivo approaches, we revealed that CD20-TCB is inducing strong and stable synapses between human T cell and tumor cells, which are dependent on the dose of CD20-TCB and on LFA-1 activity but not on FAS-L. Moreover, despite CD20-TCB being a large molecule (194.342 kDa), we observed that intra-tumor CD20-TCB-mediated human T cell-tumor cell synapses occur within 1 hour upon CD20-TCB administration. These tight interactions, observed for at least 72 hours post TCB administration, result in tumor cell cytotoxicity, resident T cell proliferation and peripheral blood T cell recruitment into tumor. By blocking the IFNγ-CXCL10 axis, the recruitment of peripheral T cells was abrogated, partially affecting the efficacy of CD20-TCB treatment which rely only on resident T cell proliferation. Altogether these data reveal that CD20-TCB’s anti-tumor activity relies on a triple effect: i) fast formation of stable T cell-tumor cell synapses which induce tumor cytotoxicity and cytokine production, ii) resident T cell proliferation and iii) recruitment of fresh peripheral T cells to the tumor core to allow a positive enhancement of the anti-tumor effect.
in vitro IFNγ neutralization
Mitson-Salazar, A., et al. (2015). "Hematopoietic prostaglandin D synthase defines a proeosinophilic pathogenic effector human T2 cell subpopulation with enhanced function" J Allergy Clin Immunol. pii : S0091-6749(15)01186-0. PubMed
BACKGROUND: IL-5+ pathogenic effector TH2 (peTH2) cells are a TH2 cell subpopulation with enhanced proinflammatory function that has largely been characterized in murine models of allergic inflammation. OBJECTIVE: We sought to identify phenotype markers for human peTH2 cells and characterize their function in patients with allergic eosinophilic inflammatory diseases. METHODS: Patients with eosinophilic gastrointestinal disease (EGID), patients with atopic dermatitis (AD), and nonatopic healthy control (NA) subjects were enrolled. peTH2 and conventional TH2 (cTH2) cell phenotype, function, and cytokine production were analyzed by using flow cytometry. Confirmatory gene expression was measured by using quantitative RT-PCR. Prostaglandin D2 levels were measured with ELISA. Gut TH2 cells were obtained by means of esophagogastroduodenoscopy. RESULTS: peTH2 cells were identified as chemoattractant receptor-homologous molecule expressed on TH2 cells-positive (CRTH2+), hematopoietic prostaglandin D synthase-positive CD161hi CD4 T cells. peTH2 cells expressed significantly greater IL-5 and IL-13 than did hematopoietic prostaglandin D synthase-negative and CD161- cTH2 cells. peTH2 cells were highly correlated with blood eosinophilia (r = 0.78-0.98) and were present in 30- to 40-fold greater numbers in subjects with EGID and those with AD versus NA subjects. Relative to cTH2 cells, peTH2 cells preferentially expressed receptors for thymic stromal lymphopoietin, IL-25, and IL-33 and demonstrated greater responsiveness to these innate pro-TH2 cytokines. peTH2 but not cTH2 cells produced prostaglandin D2. In patients with EGID and those with AD, peTH2 cells expressed gut- and skin-homing receptors, respectively. There were significantly greater numbers of peTH2 cells in gut tissue from patients with EGID versus NA subjects. CONCLUSION: peTH2 cells are the primary functional proinflammatory human TH2 cell subpopulation underlying allergic eosinophilic inflammation. The unambiguous phenotypic identification of human peTH2 cells provides a powerful tool to track these cells in future pathogenesis studies and clinical trials.